
Men, beware…are you taking this?
—-Important Message From Our Sponsor—-
“My wife can’t believe how much bigger & stiffer it is” (video proof)
I have to admit this one took me by surprise…
And by “surprise,” I mean “My jaw dropped to the floor when I finally saw it”…
Look, if you have ANY desire to get a longer, thicker, and stronger tool…
…the kind that makes women STARE in wide-eyed wonder and lick their lips…

Then you NEED to hear this guy out…
His name’s Earl and he’s far from your typical “guru.” Actually, he’s from deep in the woods…
But he’s the real deal. I’ve seen the documentation, and heard from some guys who have tried his “Maximizer” method…
And all I can say is wow, just wow…
———-
This popular remedy may cause your bones to break
So-called “proton-pump inhibitors” (PPIs) have become very popular treatments for reducing stomach acid.
These treatments, now available over the counter, include Prilosec (omeprazole), Prevacid (lansoprazole), and Noltec (ilaprazole).
There is good evidence that PPIs increase fracture risk and induce osteomalacia (softening of the bones).
This is actually what you’d expect, based on classic animal studies.
Yet newer human studies using the more soluble forms – e.g. calcium citrate and calcium gluconate – seem intentionally designed to obscure this fact.
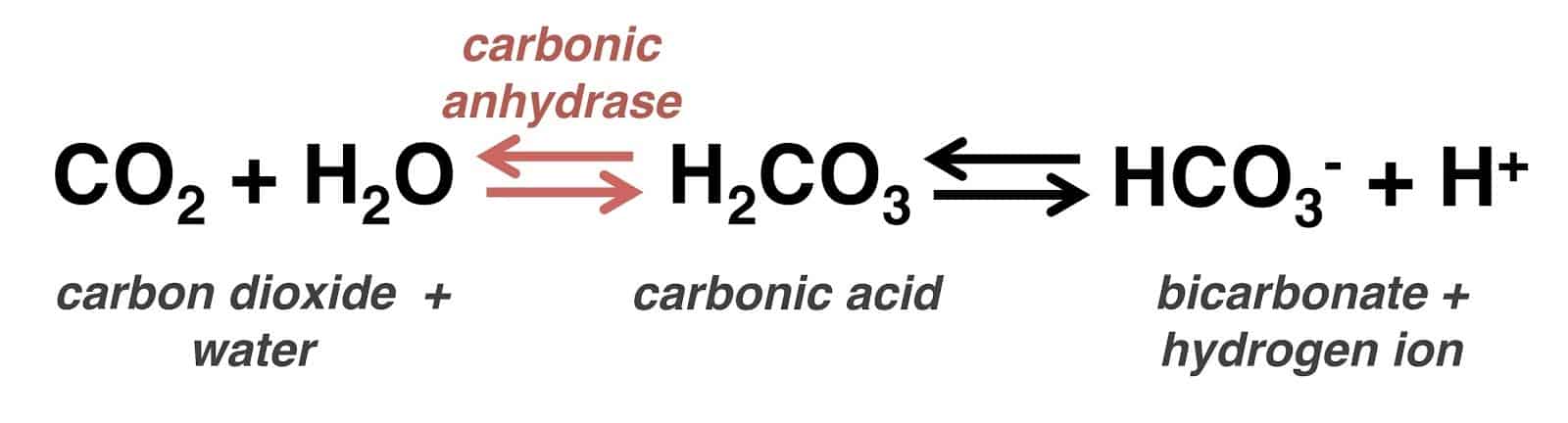
Omeprazole has also been shown to inhibit carbonic anhydrase, perhaps the real reason it inhibits stomach acid.
Carbonic anhydrase is a very important enzyme – it’s responsible for phosphate uptake in the kidneys, which it does by acidifying the urine.
Yet, on account of what’s viewed as “inconsistent and paradoxical data,” the solid link between PPIs and osteoporosis has become tainted by doubt.
However, a thorough analysis reveals that this link is real.
It shows that stomach acid influences the absorption of some forms of calcium, such as calcium carbonate and calcium phosphate salts.
Even though omeprazole actually reduces bone dissolution in certain types of lab studies, its distribution is such that it never reaches the concentrations needed for that one effect.
Omeprazole also increases some markers of bone turnover in other ways, likely through renal phosphate wasting and reduced calcium absorption.
Even though some carbonic anhydrase inhibitors can be beneficial at times, it’s always good to know when you are actually taking one.
Using a carbonic acid inhibitor can lead to problems. And sometimes patients and their doctors don’t know that the prescribed drug is a carbonic acid inhibitor.
This link to osteoporosis was first reported only a decade ago.
But enough studies have compiled since then to warrant a thorough meta-analysis on the topic:

This meta-analysis included ten studies comprising a total of 62,210 users of PPIs – and over a million control subjects.
In addition, it also examined the link to histaminereceptor antagonists (H2 blockers) using eight studies comprising 58,334 users in total.
Before PPIs were the mainstay, H2 blockers were commonly used to reduce stomach acid.
Of all these people in the studies, they found a significant association, albeit a modest one:
The risk ratio of PPI use versus spine fracture was 1.56.
And the risk ratio of PPI use versus hip fracture was 1.30.
This means an additional ~56% and ~30% chance of breaking these bones while taking omeprazole.

The association between hip fracture and H2 blockers was much smaller, however, and not statistically significant (1.12).
PPIs are more effective at reducing stomach acidity and, thus, a greater correlation could be expected.
But these two drug classes have radically different mechanisms of action.
PPIs appear to be inhibitors of the enzyme carbonic anhydrase.
Carbonic anhydrase is the main pH-regulating enzyme in the body, and its inhibition by has actually been proven:

Although the title indicates that omeprazole inhibits hydrogen potassium ATPase (H+/K+-ATPase – an enzyme that acidifies the stomach), this has always been just a theoretical assumption.
Omeprazole was developed based on its ability to inhibit gastric acid secretion, not for its binding to H+/K+-ATPase.
Carbonic anhydrase had been known for nearly a century to be a driving factor in gastric acid secretion.
And we’ve known for even longer that carbon dioxide, the enzyme’s substrate, increases stomach acidity.
“Evidence that there is a relation between blood carbon dioxide and gastric secretion is present in the work of Delhougne [1927] who found decreased acidity in response to a test meal after hyperventilation…”
“…and that of Bakaltshuk [1928] who found increased gastric acidity after inhalation of carbon dioxide…”
…”Apperly & Crabtree [1931] found that the total acid secreted varies with the blood carbon dioxide.”
Scientists have shown that carbonic anhydrase exists in gastric parietal cells (Spicer, 1978), and that inhibitors of this enzyme reduce stomach acidity (Janowitz, 1952).
Theoretically, carbonic anhydrase would be a better candidate for producing gastric acidity.
It is the fastest enzyme known with a catalytic rate of 620,000 per second, in the forward direction.
The much slower hydrogen potassium ATPase has a rate of only 1.97 per second.
So, assuming unlimited substrates, carbonic anhydrase can produce 315,000 times more protons per second than can hydrogen potassium ATPase.
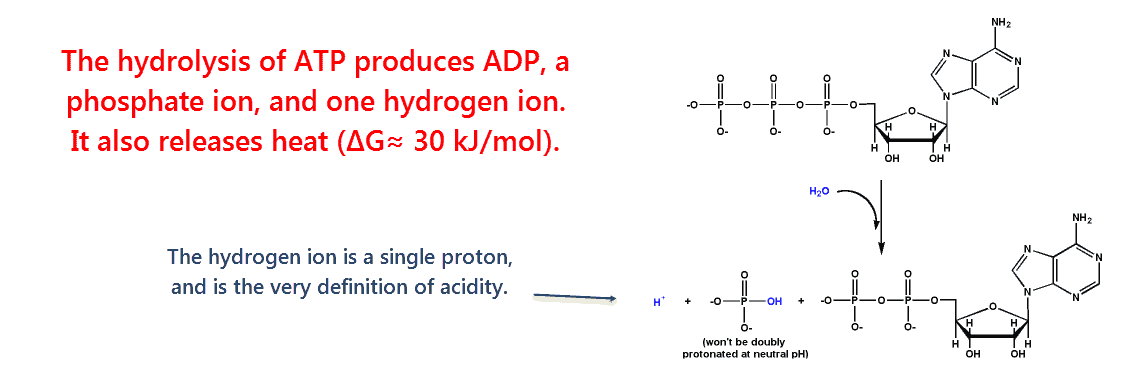
Although the hydrolysis of adenosine triphosphate (ATP) produces acidity, this is a metabolically expensive process for our bodies.
All cells have ATP hydrolase activity on the membrane to drive heat out of the cell.
But this occurs so slowly that the extracellular fluid normally remains at a basic pH.
This study found that omeprazole inhibits carbonic anhydrase in lab containers.
They found capable of exerting a 69% inhibition of the stomach isoform carbonic anhydrase-IV – at a concentration of 10 millimolars.
This effect was pH-dependent – omeprazole inhibited carbonic anhydrase in proportion to level of acidity.
This confirms why omeprazole appears to be selective for the stomach, as shown by autoradiography.
Here is one hypothesis: When omeprazole becomes ionized, it becomes less water-soluble, and hence increases its affinity for the tissues.
“The increase in activity of omeprazole to inhibit carbonic anhydrase-I, II, and IV is proportional to the reduction in pH.”
In this study, they also injected humans with 40 milligrams of omeprazole, the standard dose.
Afterwards, they measured blood and stomach carbonic anhydrase activity.
Here’s what they found:
- 64% inhibition of carbonic anhydrase-I in erythrocytes (red blood cells)
- 62% inhibition of carbonic anhydrase-II in erythrocytes
- 58% inhibition of carbonic anhydrase-IV in gastric parietal cells

This effect can easily explain all the drug’s activity in the stomach, and also hints towards its established link to osteoporosis.
Carbonic anhydrase inhibitors can either accelerate bone dissolution or inhibit it, depending on the drug’s disposition and enzyme isotype specificity.
In lab dishes, inhibiting osteoclast carbonic anhydrase-II in osteoclasts (bone cells that break down bone tissue) can prevent bone dissolution.
But inhibiting carbonic anhydrase in the kidneys can lead to urinary phosphate loss and osteomalacia.
Making things even more complicated to understand, carbonic anhydrase is a bidirectional enzyme…
Therefore, inhibiting this enzyme in erythrocytes (red blood cells) can cause metabolic acidosis by preventing alkalization.
Since it seems that carbonic anhydrase (rather than hydrogen potassium ATPase) is responsible for creating stomach acidity, inhibiting the enzyme there can inhibit calcium and magnesium absorption.
This has been known for decades:

This classic study used low-acid-secreting rats, normal rats, and four different test diets to examine this effect.
They kept the calcium content constant throughout the trials, but they administered it in four different forms:
- Calcium carbonate
- Tricalcium phosphate
- Calcium chloride
- Calcium gluconate
They tested calcium absorption every few days.
And after three weeks they sacrificed the rats and tested their bone mineral content, bone size, and femur strength.
“Bone demineralization frequently occurs following removal of acid-secreting tissue from the stomachs of man and animals.”
They found that normal rats absorbed more or less the same amount of calcium irrespective of calcium type – from 26.4% as calcium chloride to 40.7% as calcium gluconate.
Calcium absorption was predictably correlated with bone calcium content and strength, neither of which fluctuated much in the control rats.
On the other hand, calcium absorption in the low-acid rats varied substantially depending on calcium type.
Relative to the normal group, the low-acid rats absorbed:
- 37% as much calcium carbonate
- 70% as much calcium chloride
- 74% as much calcium gluconate
The relative absorption of tricalcium phosphate could not be calculated because it was negative.
Control rats absorbed 40.4% of the tricalcium phosphate that was added to their test diets.
But the low-acid rats absorbed none – while excreting some from their bones.
And, like the control rats, bone mineralization was proportional to calcium absorption – and so was femur strength.
“In rats, hypochlorhydria [low stomach acid] is associated with impaired calcium absorption, decreased bone mineral content, and decreased bone strength. This suggests that gastric acid is necessary to convert dietary calcium into a soluble form that is available for absorption.”
This effect was confirmed in other animal studies and later in human studies.
We know from some of those studies that normal men absorb calcium carbonate and calcium citrate to about the same degree (24.3% vs 22.5%)…
But low-acid men could only effectively absorb the latter type.
So why do the newer studies on calcium absorption and omeprazole always use soluble forms?
It just looks like their intent is to confuse its link to osteoporosis.
For instance, an omeprazole-calcium study published in 2010 used orange juice fortified with calcium citrate.
And it seems suspicious, considering the classic studies, that for this one they chose the most absorbable form of calcium, and in a high-acidity juice with added vitamin D.
Even though the patent of omeprazole expired before that study was conducted, the manufacturers would still potentially have to worry about liability.
But perfect absorption isn’t entirely necessary, because most Americans consume more calcium than they need.
And, since H2 blockers lower stomach acidity without causing osteoporosis, you might think another effect of PPIs could be contributing to this specific link:

For every five calcium ions in bones, we also need three phosphate ions to mineralize it properly.
This study analyzed the urinary excretion of both ions after administering acetazolamide, the most well-known carbonic anhydrase inhibitor.
They showed that acetazolamide inhibits the reabsorption of phosphate, calcium, and sodium in the proximal tubule.
However, the latter two ions were effectively resorbed in the distal tubule leaving only phosphate with a net loss.
(These tubules are parts of the kidney that help regulate things like potassium, sodium, calcium, and pH.)
“Calcium and sodium reabsorption are inhibited in parallel, whereas phosphate is inhibited out of proportion to the other ions.”
This finding is actually intuitive, as phosphate and bicarbonate are the only ions that change their ionization state with acidity.
In mildly acidic conditions, phosphate exists as a monovalent ion (monovalent refers to its ability to combine with other atoms) – dihydrogen phosphate.
In basic (opposite of acid) conditions, it becomes doubly-charged after losing a proton.
The basic phosphate (HPO42−) has a greater negative charge and is thus repelled away from cells by negative cell membranes.
“The carbonic anhydrase inhibitor, acetazolamide, also produces a marked phosphaturia in association with a modest natriuresis.”
Calcium and magnesium, on the other hand, aren’t fundamentally modified by acidity and are resorbed through ion channels in the distal tubule of the kidney.
Dietary phosphate deficiency is rare on account of its prevalence.
However, hypophosphatemia (an electrolyte disorder) due to urinary excretion does happen and is known to lead to numerous problems – including rickets in children.
And phosphaturia (hyperexcretion of phosphate in the urine) has been correlated with reduced bone density in adults.
So you might expect a lower bone density following omeprazole, based on renal phosphate wasting and reduced calcium absorption (at least on marginal diets)…
And that is despite the drug’s ability to inhibit osteoclastic bone resorption in lab containers.
This is because omeprazole has been shown to concentrate in the stomach where it exerts most of its effects – and also in the kidneys as the drug is excreted.
It’s been shown that 80% is excreted in the urine and the remainder in the bile.
And full-body disposition studies on rats report negligible distribution in fat and muscle cells.
The plasma half-life of less than one hour testifies towards the solubility of this drug.
Acetazolamide, another drug that inhibits bone resorption in lab containers, has also been shown to induce osteomalacia in live animal studies.
Such is the complexity of carbonic anhydrase, a ubiquitous bidirectional enzyme with over ten isoforms.
So, even though the effects of omeprazole could likely be overcome through supplementation or a high-mineral diet, it’s important to know about them so you can take precautions.
The link between PPIs and osteoporosis is likely real.
And it might be due to the combined effect of reduced calcium absorption and increased dihydrogen phosphate elimination.
Inhibiting stomach acid is a bad idea for other reasons besides mineral balance, such as the risk of food allergies following incomplete proteolysis…
So you should probably do it only when you absolutely have to.
If you absolutely must inhibit stomach acidity for some reason, such as to heal an ulcer, then you could alternate omeprazole with the safer H2 blockers or take it with them in lower doses.
Limonene has no known side effects and has been shown to pretty much eliminate heartburn in most people.
Although its precise mechanism of action isn’t officially known, there is reason to believe that d-limonene inhibits acid-sensing TRPA1 receptors in the esophagus.
So even though d-limonene doesn’t appear to influence gastric acidity at all, it is uniquely capable of blocking the perception of heartburn at the receptor level.
—-Important Message for Men Taking Blood Pressure Treatments—-
WARNING: Are you taking any of these?

———-
