
[cmamad id=”25538″ align=”center” tabid=”display-desktop” mobid=”display-desktop” stg=””]
Studies show this natural immune component destroys cancer cells… So why is it outlawed?
—-Important Message—-
Doctors using GcMAF to cure cancer patients found dead under mysterious circumstances

Listen… This goes way deeper than I ever imagined…
But I can’t tell you about it here… Email isn’t safe anymore…
Here’s what’s happening to doctors and health researchers who go anywhere near GcMAF…
———-
This Natural Treatment Fights Cancer Better Than Chemo
Few things are more notorious than GcMAF in the realm of alternative and outlawed anticancer therapies…
Yet is it really worth the expense to source for a cancer patient?
Perhaps it’s not…
There are many other alternative remedies that are cheaper that people can buy online.
Nonetheless, GcMAF is certainly more effective than chemo and far safer – and is certainly a better “alternative” to radiation.
Yet if GcMAF works exactly as it appears to, then there are other natural things that can do the same at a fraction of the expense.
In other words, there could be better alternatives in the pool of “alternatives” to choose from.
Many other natural supplements have a more established track record and can be taken orally – unlike GcMAF, which doesn’t survive digestion.
This is because GcMAF appears to work mainly as an activator of macrophages, no more/no less – the function it was partially named after.
The other half of its name, the “Gc” attribute, refers to the historical name of vitamin D binding protein (VDBP).
The vitamin D binding protein was originally named “group-specific component of serum.”
And then it was subsequently referred to as “Gc-globulin” for short.
Yet these two proteins are one and the same.
And VDBP/Gc has other functions besides…
In addition to transporting vitamin D around in the body and delivering it to tissues, it also has a high affinity binding domain for actin.
This function is also important.
Actin is a muscle protein released into the body during cachexia.
(Cachexia = weakness and wasting of the body due to severe chronic illness.)
It has been shown that too much actin in the plasma can be fatal if allowed to self-associate, polymerize, and then inhibit blood flow.
So inhibitors of this process have evolved.
The vitamin D binding protein, along with gelsolin, usually prevents this from happening.
Yet death can still occur should their actin-binding capacity become overwhelmed.
There is a twenty-fold higher concentration of circulating VDBP than vitamin D itself (its eponymous ligand)…
So you could consider actin-binding to be its primary function…
…especially when considering its very high affinity for actin.
Yet the multifunctional Gc-protein has another function.
It was discovered incidentally by Nobuto Yamamoto et al. over a quarter century ago.
Yamamoto discovered that treating mouse cells with lysophosphatidylcholine increased phagocytic activity of macrophages…
But only when the cells were incubated with fetal calf serum.
Yamamoto fractionated, electrophoresed, and immunostained the calf serum…
And then he discovered that Gc-protein was the only serum component indispensable for macrophage activation.
He showed in later experiments that Gc-protein is activated by an enzyme on B-cells called beta-galactosidase.
This membrane component removes the galactose from Gc-protein.
This can be induced by lysophosphatidylcholine, neatly explaining two of Yamamoto’s previous findings.
And after diffusing towards a second enzyme (sialidase on T-cells), the Gc-protein becomes fully activated and is now termed GcMAF.
(Again, that is short for “Gc–macrophage activating factor.”)
This protein is real and slightly different from normal Gc-protein…
And its ability to stimulate macrophages has been confirmed by others.
The vitamin D binding protein also binds fatty acids.
It even activates neutrophils through a different part of the protein than its macrophage activation domain.
Most other claims have also been independently confirmed…
…such as the ability of nagalase to inactivate GcMAF.
The enzyme nagalase is found elevated in patient’s serum during progressive stages of cancer.
But increased levels can be detected within the tumor micro-environment at any stage.

Nagalase can do this because it’s an N-acetylgalactosaminidase.
(That’s an enzyme with the function of removing terminal N-acetylgalactosamine units from proteins and polysaccharides.)
After first being primed by B- and T-cells, the fully-activate GcMAF still has one N-acetylgalactosamine remaining.
So for the Gc-protein to become active GcMAF it must:
- Have its sialic acid removed by activated B-cells
- Have its galactose removed by T-cells
- Dodge nagalase, which can remove its remaining N-acetylgalactosamine
Macrophages and immature dendritic cells have a well-characterized C-type receptor that exclusively binds N-acetylgalactosamine.
And this probably explains why macrophages are most affected by GcMAF.
“One of the C-type lectin receptors exclusively expressed by dendritic cells and macrophages is the macrophage galactose-type lectin (MGL).”
The ability of GcMAF to bind and activate macrophages makes complete sense once we understand two things:
- That macrophages also have a considerable amount of actin on the cell membrane (Hartwig, 1989)
- That Gc-protein strongly binds this (McLeod, 1988)
Macrophages use actin for motility and engulfment. Actin is also responsible for the shape changes seen in platelets upon activation.
Activation of the C-type receptor using anything bearing a terminal N-acetylgalactosamine induces tumor necrosis factor alpha (TNF-α), a cytokine named for its ability to inhibit cancer.
Besides increasing macrophage activity and superoxide production, GcMAF may also inhibit cancer by inducing TNF-α.
So the origin of GcMAF may initially seem confusing. And it may have a reputation for doubtful efficacy due to bad press by the AMA.
But, actually, nearly every facet of its function has been independently confirmed.
This is very important because the lion’s share of all GcMAF studies have been published by its discoverer, Nobuto Yamamoto – a person who also holds patents on GcMAF.
After GcMAF got popular enough to threaten profits for the AMA and Big Pharma, they went over Yamamoto’s studies with a fine-toothed comb.
They compiled enough apparent irregularities to have a few Yamamoto articles (coincidentally the ones about treating humans) retracted by journal editors seven years after initial publication.
This would never happen to any article about a Merck, Pfizer, or Eli Lilly product – even if it was riddled with the most egregious errors.
Yet it must be admitted that a few Yamamoto articles do have some minor irregularities…
For example, Yamamoto states in one 2007 article that “Administration of 100 nanogram GcMAF to humans results in the maximal activation of macrophages with 30-fold increased ingestion index and 15-fold increased superoxide-generating capacity.”
But then he cites an animal study!
This inaccuracy is listed as the first main point in the critique published in Cancer Immunology, Immunotherapy, the same journal that published the Yamamoto article under review.
[cmamad id=”25539″ align=”center” tabid=”display-desktop” mobid=”display-desktop” stg=””]
Since this letter to the editor directly preceded the retractions, we can infer that it initiated the retractions.
Yet a deeper investigation unearths that this was probably an innocent error..
Yamamoto stated earlier in US patent #5620846A that “Administration of a minute amount (10 pg/mouse; 100 ng/human) of GcMAF resulted in a greatly enhanced phagocytic capacity of macrophages.”
This statement comes in various forms throughout his articles…
And it seems that he just worded it incorrectly in the 2007 article.
One hundred nanograms is the mouse dose extrapolated to humans. And the 2007 article is the only one that left that out.
Nobuto Yamamoto was 82 years old when he wrote that sentence…
And, as a Japanese native raised before WWII, he was not taught English at a young age.
The second and third points raised are dubious – essentially just pointless bitching about minor semantics.
Such errors can be found in just about any article written by a scientist not in full command of the English language.
In my view, a worse error is one by David Noakes, the former owner of a GcMAF company. He states (2013):
“In serum, nagalase acts as an endo- (but not as an exo-) enzyme, being unable to deglycosylate an N-acetylgalactosamine residue of GcMAF.”
But in fact, the exact opposite has been proven to be true (Saharuddin B. Mohamad, 2001):
“Although the endo-type alpha-NaGalase has been reported in bacteria, only the exo-type of alpha-NaGalase has been reported in humans.”
“Tumor-derived alpha-NaGalase having higher activity than normal alpha-NaGalase, had higher substrate specificity to the exo-type substrate than to the endo-type substrate, and still maintained its activity at pH 7.”
An endo-type galactosidase will cleave the sugar in the middle of a polysaccharide chain, while an exo-type will only cleave those found at the very end.
David Noakes et. al. need nagalase to be an endo-enzyme or they’re faced with this difficult question:
“How can such low doses of GcMAF be effective in the presence of high nagalase activity (which inactivates it) as is observed in advanced cancer patients?”
So, in spite of all the data actually proving the opposite, Noakes decides to reference an offhand speculation in a Yamamoto article as proof.
But nagalase doesn’t need to be an endo-enzyme to resolve the paradox anyway because of its low activity at serum pH of 7.4.
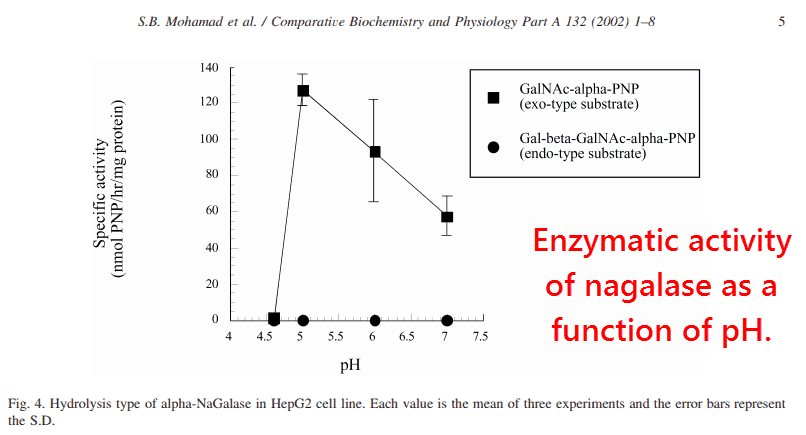
Nagalase is normally found within the lysosomes (pH ≈ 5), an acidic compartment that breaks down and internally recycles cell components.
Moreover, nagalase isn’t even increased all that much in cancer – not even doubled.
Moreover, VDBP/Gc is cleared by the liver with a plasma half-life of 2.5 days.
So you wouldn’t necessarily expect nagalase to deactivate all GcMAF before it is removed from circulation or becomes attached to macrophages.
Macrophages have a higher affinity for GcMAF than nagalase – so can be assumed to be the prime target.
Business owners such as Noakes have much to gain by speaking favorably about their products.
If for no other reason, the discoverers of new molecules also tend to be overly optimistic to maintain their legacy and justify their life’s work.
The discoverer of statins, Akira Endo, still promotes them as if they were a gift of nature.
Despite all of the evidence proving that cholesterol isn’t pathological and statins actually kill people – partially by inhibiting other isoprenes such as Coenzyme Q10 – Endo still writes at the age of 75 self-congratulatory articles such as this one: “A gift from nature: the birth of the statins.”
Yet despite the many over-enthusiastic viewpoints surrounding GcMAF, it has been confirmed to work by unaffiliated academic scientists.
Nagalase is in fact a biomarker of cancer, despite what the critics say – and especially the higher grades.
And GcMAF has been shown to stimulate macrophages by numerous study groups.
Yet, you don’t need such an esoteric product to stimulate macrophages through C-type lectin receptors (MGL).
Just about any molecule ending with N-acetylgalactosamine will do. And some have been used for cancer before.
Endotoxin stimulates macrophages. And its active O-antigen component always contains an N-acetylgalactosamine.
Aloeride (a polysaccharide in Aloe vera) does the same.
Acemannan is another polysaccharide derived from Aloe vera. And it too has immuno-stimulating properties and an N-acetylgalactosamine.
Yet there are few people out there promoting Aloe vera… Nobody can make money off a plant you can legally grow at home.
Since nobody trumpets it, this cactus plant is left alone by the AMA and Big Pharma companies…
Yet if it were to became immensely popular, they would surely find ways to discourage its use.
In fact, N-acetylgalactosamine itself has been shown to prime macrophages.
There appears to be nothing special about GcMAF in this regard – besides unusually high affinity.
And that may be a consequence of the actin-binding domain lying adjacent to its serine-linked N-acetylgalactosamine.
Endotoxin has been shown to activate macrophages and release TNF-α…
It has been used to prime the immune system against cancer for over a century.
Doctor William Coley was the first to report that heat-inactivated bacterial cell lysates (which likely contained endotoxin or something similar) would induce the autolysis of tumors.
This, of course, made Coley a target of other medical doctors…
Yet “Coley’s toxins” have been used by a minority ever since.
So, even though GcMAF is completely safe and probably works, at least to some extent, it may not be THE magic bullet as is often claimed by its manufacturers.
It may not be worth boarding a plane to Germany and paying a considerable amount for a product that, in essence, has effects indistinguishable from other products that are easier to get.
And nagalase is a kitten compared to hyaluronidase, a more powerful cancer enzyme designed for the extracellular space and active at neutral pH.
Hyaluronidase is an endoglycosidase that turns hyaluronic acid into confetti, thereby removing contact inhibition for cancer growth and creating space for metastasis.
—-Important Message for Men Who Want to Boost Member Size—-
An easy, natural way to grow a bigger penis
Most men wonder: “Can I really increase my penis size?”
And the answer is yes. Yes, you can.
Most men assume they have to use some weird pumps or take dangerous Big Pharma treatments just to make their penis a little bit bigger…
But not anymore. There’s a better, natural way to get a bigger and fatter penis.
And it really works – by naturally increasing important male hormones such as testosterone and DHT.
You just paint this natural mineral on your penis and watch it grow…

———-

- Coley, William B. "II. Contribution to the knowledge of sarcoma." Annals of surgery (1891)
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1428624/pdf/annsurg01156-0040.pdf - Pugh, Nirmal. "Characterization of Aloeride, a new high-molecular-weight polysaccharide from Aloe vera with potent immunostimulatory activity." Journal of agricultural and food chemistry (2001)
http://www.psorioderm.com/public/file/cikk/aloe-vera-psoriasis-study3.pdf - Mohamad, Saharuddin. "Tumor cell alpha-N-acetylgalactosaminidase activity and its involvement in GcMAF-related macrophage activation." Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology (2002)" Annals of surgery (1891)
https://www.saisei-mirai.or.jp/gan/pdf/2002-tumor-cell-alpha-N-acetylgalactosaminidase-activity-gcmaf-macrophage-activation.pdf - Iida, Shin-ichiro. "Interaction of Human Macrophage C-type Lectin with O-Linked N-Acetylgalactosamine Residues on Mucin Glycopeptides." Journal of Biological Chemistry (1999)" Annals of surgery (1891)
http://www.jbc.org/content/274/16/10697.full.pdf
- Promising role for Gc-MAF in cancer immunotherapy: from bench to bedside
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5686300/ - GcMAF as a Cancer Treatment
https://www.healthline.com/health/gcmaf-cancer-treament
