
[cmamad id=”21378″ align=”center” tabid=”display-desktop” mobid=”display-desktop” stg=””]
Doctors say these men eat a “poor” diet, yet their hearts are healthier than any others in the world… Here’s why…
—-Important Message—-
These 5 Foods Open up the Blood Vessels for Better Erections
Most men do not know that the true cause of high blood pressure is narrow blood vessels.
You see, when blood vessels get narrow and closed off, blood flow suffers…
And this raises blood pressure because the body is working harder to get good blood flow:
And men NEED good blood flow to get great erections.
The more blood is flowing into the penile chambers, the stiffer and bigger his erections can be…
This is why men with high blood pressure tend to experience erectile difficulties.
But I’ve recently discovered these 5 simple foods that open up the blood vessels – even in the penis…
————
The shocking reason why men in Greenland never have heart attacks
Ancel Keys said that cholesterol causes heart disease.
But today we know that isn’t so.
Today, new “risk factors” keep proving Ancel Keys wrong.
And yet followers of this antiquated 1950s science have not gotten the memo.
“These findings suggest that other important risk factors remain to be identified.”
The more reliable and realistic evidence strongly suggests that, with the proper fatty acid ratio and ascorbate intake (for a high collagen turnover), there will be no cholesterol deposits in the arteries and no ischemic events.
Evidence implicating estradiol in heart attack supports this conclusion, as does every other piece of evidence I’ve ever come across.
[cmamad id=”21379″ align=”center” tabid=”display-desktop” mobid=”display-desktop” stg=””]
You might be wondering: “What does estradiol have to do with ascorbate?”
Nothing, really… But that’s not the issue in this case…
Heart attack is just one specific subtype of “cardiovascular disease.”
And it has more to do with thrombosis (blood clots) than with atherosclerosis (hardening of the arteries).
Yes, it’s true – a heart attack is more similar to blood clots and stroke than it is to atherosclerosis.
A heart attack is basically what happens when a blood clot blocks the oxygen supply to the heart.
Even though the most convincing mechanism behind atherosclerosis is low ascorbate intake, the most consistent etiological relation for heart attack is omega−6 fatty acids.
This is because the shorter omega−6 fatty acids can elongate into arachidonic acid, the direct precursor for all 4-series leukotrienes and 2-series prostaglandins.
Of the latter class of eicosanoids (the prostaglandins), thromboxane A2 is a notorious platelet aggregator.
Thromboxanes are synthesized by cyclooxygenase, so they are technically prostaglandins.
Cyclooxygenase inhibitors, such as aspirin, increase bleeding time and inhibit stroke by lowering thromboxane A2.
Some of the best epidemiological evidence for comes from the Inuit of Greenland, a population entirely free of ischemic cardiovascular disease.
This occurs despite consuming an essentially an all-meat diet, another nail-in-the-coffin for the simplistic Ancel Keys’ naïve cholesterol hypothesis.
“Myocardial infarction commonly occurs in patients with none of the major known risk factors for the disorder. This suggests that important risk factors remain to be discovered.”
The reason for this is simple: The Greenland Inuit consume practically no omega−6 fatty acids at all.
Marine oils contain nearly all omega−3 fatty acids… And, despite the issues with PUFAs in general, these don’t cause heart attack and stroke.
What you instead find in the Inuit is thromboxane A3, and not the blood-clotting thromboxane A2.
Thromboxane A3 is formed from eicosapentaenoic acid, a long-chained omega−3 fatty acid, not the elongated omega−6 fatty acids derived from temperate seed oils.
Besides a near-complete absence of heart attack and stroke, the Greenland Inuit also have bleeding times twice as long as their Danish neighbors.
And quite surprisingly, aspirin actually shortens bleeding time in the Inuit – that’s called the “reverse effect.”
Apparently, it does this by inhibiting cyclooxygenase and hence thromboxane A3 synthesis.
No situation in biology better highlights the difference between the two lipid types.
Thromboxane A2 is such a powerful pro-thrombotic agent that it’d been named for it…
Yet thromboxane A3 – the omega−3 analog – has a diametrically opposite effect.
So, even though all polyunsaturated fatty acids increase peroxidation and lipofuscin formation and should be limited, it is only the omega−6 class that induces ischemic cardiovascular disease.
But what about estrogen’s involvement in this?
Unsaturated fatty acids have consistently been shown to displace estrogen from its serum-binding proteins at physiological concentrations.
So you would expect an increase of unsaturated fatty acids to increase free estradiol – which can be seen more as a biomarker than the actual cause.
Free estradiol could be a proxy for the arachidonic acid that displaces it.
Now the evidence, which is very consistent – and convincing:

In this study, they used 61 heart attack victims who were perfectly age matched against 61 control subjects.
“Of the 15 highest estradiol values, 13 occurred in subjects with coronary disease.”
This was a spin-off of one of the longest-running studies to date: The Framingham Heart Study that was initiated in 1948 and is still running to this day.
Lead author Gerald Phillips noted significantly increased concentrations of estradiol in heart attack victims…
But with absolutely no consistent changes in DHT or testosterone.
This is what you’d expect from high omega−6 intake, because arachidonic acid only displaces estrogen binding, not androgen binding.
Saturated fatty acids are relatively inert in this respect.
“The mean serum estradiol level was significantly higher in the subjects with coronary disease… The mean serum testosterone level was not significantly different.”
Dr. Phillips published two more articles on this topic, both before this study… He did this study to further demonstrate the point.
And why not? This is an important finding.
Scientists and doctors must know when their theories have gaping holes in them.
Below is the first demonstration published, which was done on an entirely different set of patients and controls:

In this study, Phillips took 15 of his heart attack patients and age matched them to control subjects.
Back in 1976, he probably did not think to check estradiol levels because there was no precedent – yet 87% of these heart attack patients had an obvious estrogenic appearance.
“Seven of the patients had a strikingly slow rate of beard growth, three had evidence of gynecomastia [man boobs], and three had a loss of libido.”
Because of these observations, perhaps especially the man boobs, he drew blood…
And he found elevated estradiol levels in the heart attack patients.
He noted a 30% increase in circulating estradiol concentrations and a 35% increase in estrone.
As with his later Framingham study, the androgens were not significantly different between the two groups:
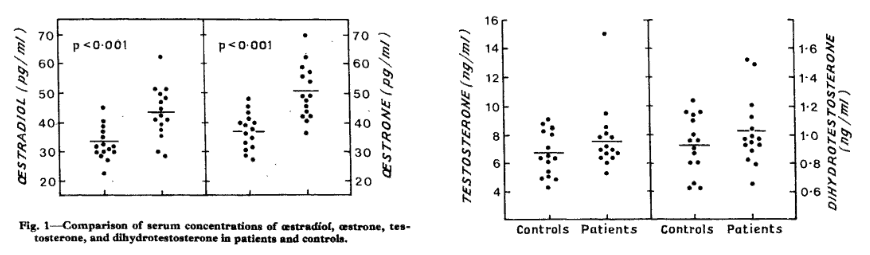
And this is a consistent finding…
Besides Dr. Phillips’ multiple studies demonstrating this effect, it was confirmed by Dr. Myron Luria in 1981 and again by Dr. Edward Klaiber in 1982.
“The slow beard growth and decreased libido, and possibly the gynecomastia, preceded the myocardial infarction.”
The 1982 Klaiber study is especially convincing because it compared estradiol levels near the time of the heart attack – on the very same day.
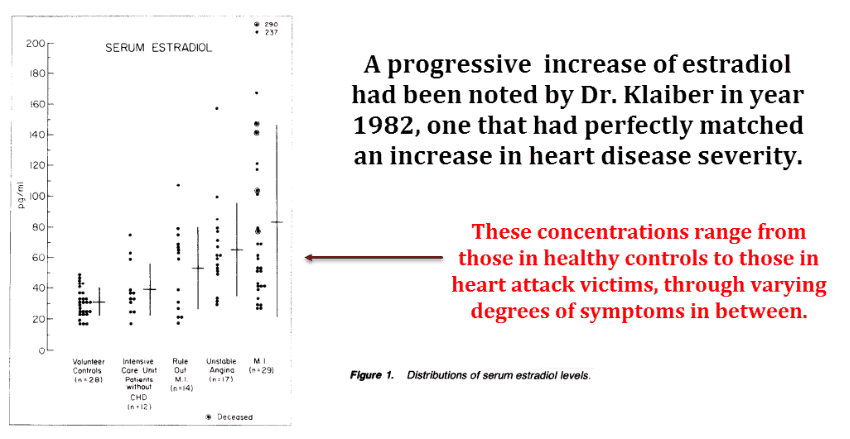
This article shows a larger estradiol charge during the acute phase of the heart attack…
And that suggests in the studies by Phillips those levels were allowed to drop towards baseline after the convalescent patients reduced food intake – most specifically omega−6 intake.
In Phillips’ study:
“Gynecomastia was not detected in any of the normal subjects.”
The Klaiber study also shows a moderate and progressive reduction of estradiol in the days that follow the heart attack.
This argues against increased aromatase expression – which, incidentally, was Klaiber’s own explanation for the increased concentrations.
So there is a consistent increase of estradiol during angina and following heart attacks…
But these symptoms also correspond to a pre-diabetic state – and that also implicates omega−6 fatty acids.
In yet another study, Dr. Phillips demonstrates this relation to insulin and glucose…
He used the same fifteen feminized patients from the other study and gave them an oral glucose load:

Doctor Phillips had noted insulin resistance in his fifteen estrogenic patients – and all of them had just had heart attacks.
This was a very significant correlation, with Pearson correlation coefficients approaching 1.0 – the perfect straight-line value.
“The ratio in serum of the concentrations of estradiol to testosterone correlated with serum glucose area (r = 0.69), insulin area (r = 0.80), and the ratio of insulin area to glucose area (r = 0.64) in the glucose tolerance test.”
As far as I know, estrogen does not directly cause insulin resistance – yet the omega−6 fatty acids that displace it can.
The involvement of omega−6 fatty acids in type 2 diabetes is a highly consistent finding…
Perhaps the best explanation is that they lead to a reduced transmembrane glucose flux:
Omega−6 fatty acids actively displace omega−3 fatty acids from the cell membrane where they reside…
And specifically DHA… in a substitution that leads to increased cholesterol incorporation in the cell membrane.
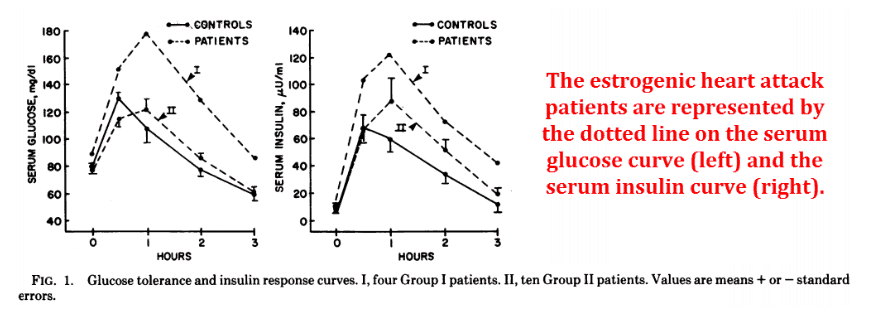
Yet this is not cholesterol’s fault, but the action of arachidonic and osbond acids.
(Those are fancy names for some omega–6 fatty acids.)
Since we make cholesterol ourselves in the liver, eating it directly has little effect on the concentrations circulating in our blood.
In fact, the less cholesterol we eat, the more we manufacture…
This homeostatic mechanism guarantees a more-or-less steady state.
Even vegetarians in India get cardiovascular disease, and they get it at greater rates than the all-meat-eating Inuit.
“Another difference between the two groups was the lower intake of fat of animal origin in the patients.”
Fatty acid correlations against diabetes are so strong that a mere presence of insulin resistance strongly suggests an elevated omega−6 to omega−3 ratio.
Since these patients had high estrogen levels and insulin resistance, I can think of only one common denominator…
Omega−6 fatty acids are perhaps the only thing shown to cause both.
This study demonstrates that very well:

Unlike the other studies above, this was an experimental biochemical study – and a rigorous one at that.
The researchers purified the sex-steroid-binding protein from human serum and used radiolabeled steroids, gel electrophoresis, and radiography to determine affinity.
And they used equilibrium dialysis for confirmation.
They found that saturated fatty acids all reliably increased steroid binding to the blood protein responsible for doing such…
Whereas unsaturated fatty acids decreased it.
And not only that, they discovered that unsaturated fatty acids displaced estradiol to a far greater degree than androgens:
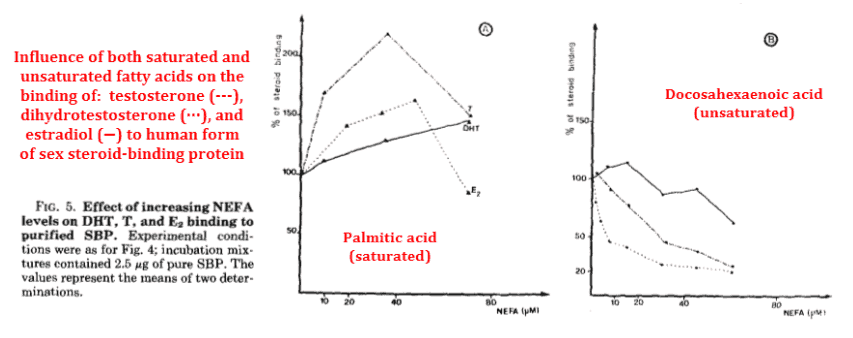
Of course, this implies that consuming unsaturated fatty acids would increase free estradiol, perhaps explaining its correlation to heart attack – as arachidonic acid can induce both.
Although they used docosahexaenoic acid (an omega−3 fatty acid) in the graph above, they also showed that the much-maligned arachidonic acid has essentially the same potency.
“Thus, unsaturated, non-esterified fatty acids in the concentration range 1-100 μM are more inhibitory for estradiol binding than for testosterone or dihydrotestosterone binding.”
Arachidonic acid is the precursor for thromboxane A2, the platelet-aggregating prostaglandin that is involved in thrombosis.
However, even though omega−3 fatty acids can displace estradiol (just like omega−6 fatty acids can), they don’t lead to ischemic cardiovascular disease – because they produce thromboxane A3, not thromboxane A2.

And the concentrations used in the study are well within the range of what’s achievable through food.
“The arachidonic acid levels can be 10 times higher than those of pregnancy in some pathological conditions characterized by major stress, under which circumstances the fatty acid/SBP ratios could be considerably higher.”
You wouldn’t expect omega−3 fatty acids to be correlated with ischemic cardiovascular disease…
And, as we can see with the Greenland Inuit and also studies of Polynesians, they are not.
Yet the long-chain omega−3 “marine oils” do cause lipofuscin, which is certainly reason enough to limit their intake.
Omega−6 fatty acids displace omega−3 fatty acids…
So eating less omega−6 allows people to maintain a better omega−3 to omega−6 ratio while consuming only trace amounts of PUFAs.
Docosahexaenoic acid (DHA) has a low turnover rate and it is very efficiently recycled.
While avoiding the omega−6 fatty acids from seed oils, the trace amounts of omega−3 fatty acids you can get from plants and their leaves, grass-fed beef, and grass-fed dairy should be all anybody needs.
“They suggest that the fatty acid content of the sex-steroid-binding protein environment may result in lower steroid hormone binding and thus increased free hormone levels.”
“Taken together, our results indicate that unsaturated fatty acids modify the binding, electrophoretic, and immunological properties of serum SBP. These modifications may occur as a result of the unsaturated fatty acids interacting with the SBP to reduce steroid accessibility and, at the same time, changing the net charge on the protein.”
——

- Martin, Marie-Elise. "Modifications of the properties of human sex steroid-binding protein by nonesterified fatty acids." Journal of Biological Chemistry (1986)
http://www.jbc.org/content/261/6/2954.full.pdf - Phillips, Gerald. "Relationship between serum sex hormones and glucose, insulin and lipid abnormalities in men with myocardial infarction." Proceedings of the National Academy of Sciences (1977)
http://www.pnas.org/content/pnas/74/4/1729.full.pdf - Phillips, Gerald. "Association of hyperestrogenemia and coronary heart disease in men in the Framingham cohort." The American journal of medicine (1983)
http://doi.org/10.1016/0002-9343(83)91078-1 - Klaiber, Edward. "Serum estrogen levels in men with acute myocardial infarction." The American journal of medicine (1982)
http://sciencedirect.com/science/article/pii/0002934382907793 - Phillips, Gerald. "Evidence for hyperestrogenemia as a risk factor for myocardial infarction in men." The Lancet (1976)
http://sciencedirect.com/science/article/pii/S0140673676929688
- The importance of the ratio of omega-6/omega-3 essential fatty acids.
https://www.ncbi.nlm.nih.gov/pubmed/12442909 - Essential Fatty Acids | Linus Pauling Institute | Oregon State University
https://lpi.oregonstate.edu/mic/other-nutrients/essential-fatty-acids

Leave a Reply