
These foods may even keep you from getting blood sugar problems years later

—-Important Message—-
Did you know there was an Englishman in the 1800’s who gave women over 2 million orgasmic releases?
The wild thing is that each one of these orgasmic relesaes was achieved by using nothing more than his pointing finger.
And it took him less than 30 seconds to pull it off… to get her shuddering and shivering with pleasure…
Don’t believe me?
See how it works right here (warning: NSFW)
———-
How to get more testosterone-boosting naringenin from food
Of all the natural antidiabetic compounds extracted from plants, none have consistently been shown to be more effective than naringenin.
Naringenin belongs to a class of citrus flavanones along with hesperitin, eriodictyol, and isosakuranetin.
In fruits, these are generally found as glycosides attached to sugar molecules, yet become aglycones within the body.
The aglycones alone are capable of penetrating the cell membrane and, considering their target, are expected to be considerably more active.
Fruits and herbs containing naringenin have been used to enhance glucose metabolism for centuries, yet its interest to Western science is rooted in the popularity of grapefruit diets for weight loss.
All citrus fruits contain naringenin, but grapefruit has more than most other popular varieties (e.g., oranges, tangerines, limes, or lemons).
Mandarins are reported to contain the most, but caution is warranted.
Clementines contain the least and are nearly identical to mandarins.
Both types are marketed as “Cuties®” or “Halos®,” the two most popular brands of small oranges sold at U.S. grocery stores.

Fortunately for us, naringenin has been very well studied, so there’s multiple review articles on the subject…one of which includes 99 citations (Hartogh, 2019)!
Consistent trends emerge from this comprehensive body of evidence.
Naringenin: (1) increases fatty acid β-oxidation, (2) clears excess plasma triglycerides, and (3) lowers blood glucose.
Besides those general observations, the findings occurring on the transcriptional level are just as consistent.
Naringenin upregulates both uncoupling proteins (UCP1, UCP2), glucose transporter-4 (GLUT4), and adiponectin, among other things.
Although adiponectin SOUNDS like something that could make you fat, the truth is actually the reverse.
Adiponectin has been shown to reverse insulin resistance (Yamouchi, 2001) and is inversely correlated with obesity (Arita, 1999).
Adiponectin has adipo- in its name — the Latin prefix for fat — not because of its effects, but because it’s made exclusively by adipose cells.
Adiponectin will make you skinny and does so through AMP-activated protein kinases.
So how can one molecule do so many things, consistently?
The answer lies in the activation of two nuclear receptors which are capable of inducing the transcription of dozens of enzymes, transporters, receptors, and various other unclassifiable proteins.
This dual activity explains the unique efficacy of naringenin.
Other similar citrus flavanones, such as hesperetin, can only activate one PPAR subtype at most.
Naringenin activates both PPAR-α AND PPAR-γ.
“The manifold effects of naringenin, include the induction of β‑oxidation and anti-inflammation, suggest an underlying mechanism, similar to the activities of PPARα and PPARγ agonists […]” ―Goldwasser, 2010
This PPAR activity of naringenin reliably translates into lower body fat, greater muscle endurance, and increased body heat:

On account of previous reports, scientists from MIT had gained a suspicion that naringenin was likely a PPAR agonist…
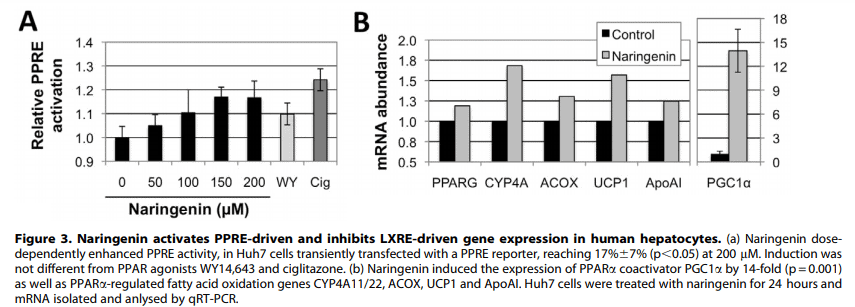
…so they added naringenin to normal liver cells and checked the result.
They found increases of uncoupling protein-1, cytochrome-4A11, apoprotein A, PPAR-γ, and acyl-coenzyme A oxidase — all of which are PPARα–inducible genes.
They spliced DNA encoding for acyl-coenzyme A oxidase to the one encoding for luciferase, an enzyme which creates detectable light.
In this way, they could visualize naringenin as it activates PPAR-α:
They also proved this more formally using a fusion protein of PPAR-γ and GAL-4, a biochemical chimera of two transcription factors linked together.
Activation of the PPAR-γ half would transcribe for genes through the GAL-4 side of the chimera, and after DNA is inserted with GAL-4 response elements you can get measurable activity from PPAR-γ ligands.
They used GAL4 because it’s an insect transcription factor and, as such, it doesn’t interact with anything besides the DNA added to the cell.
They verified that this technique works by using known synthetic activators of the PPARs, so it’s reliable despite its complicated nature.
Moreover, they confirmed that naringenin is an effective agonist of both PPAR-α and -γ.
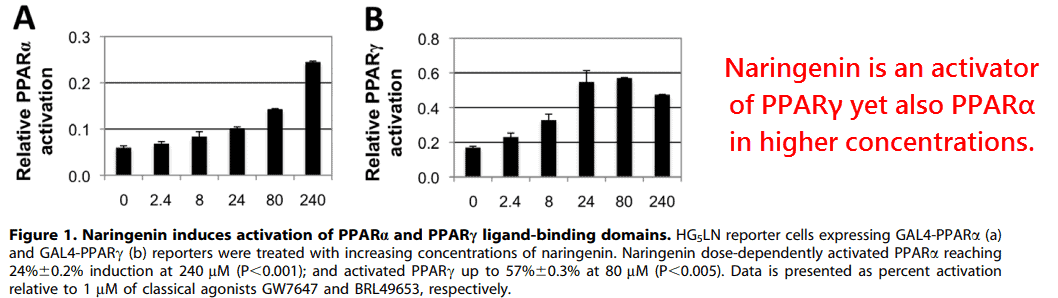
Naringenin was capable of doubling PPAR-γ transcription at modest concentrations (8μM)…
In other words, approximately what’s found in human plasma after drinking ~.6 liters of grapefruit juice (Erlund, 2000)…
But higher concentrations were required to activate PPAR-α.
Yet a multitude of studies in vivo indicate it has effects on PPAR-α at lower concentrations, or levels less than you’d expect would be effective through transcription assays and post mortem chemical distribution studies (El Mohsen, 2017).
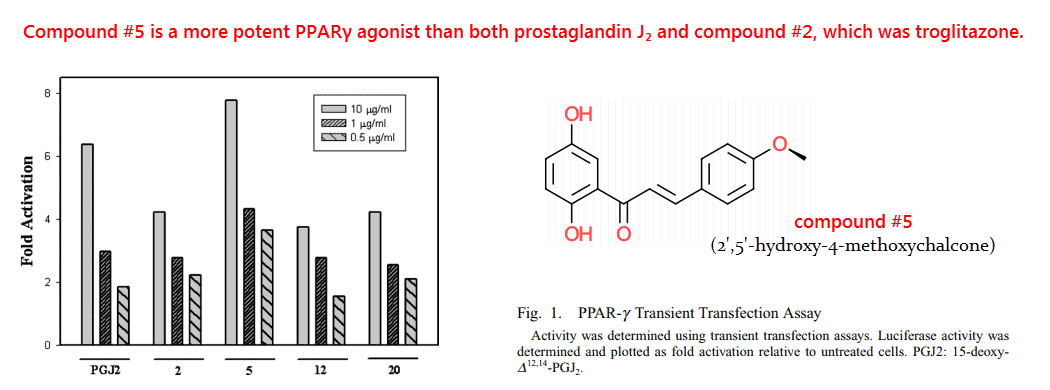
But there’s indication that metabolites of naringenin, especially its methoxychalcones, are more potent PPAR agonists than the unmodified flavanone.
For instance: 2′,5′-hydroxy-4-methoxychalcone is a more potent PPAR-γ agonist than both prostaglandin J2 and troglitazone…
…which is saying a lot considering their high affinities.
Prostaglandin J2 is the most well-known endogenous ligand of PPAR-γ and troglitazone, a thiazolidinedione, is an antidiabetic treatment.
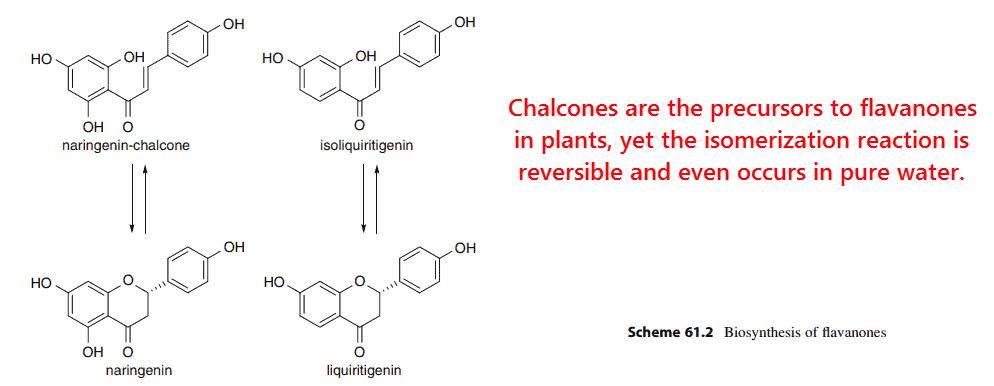
Chalcones are natural metabolites of flavanones in mammals and direct precursors for them in plants.
Flavanones and their open-ring chalcones coexist together in equilibrium (Cisak, 1992), readily transposing back-and-forth in water at basic pH.
Naringenin has been shown to upregulate apinectin (Liu, 2008), a PPARγ-inducible hormone, and so has naringenin chalcone (Horiba, 2010).
Although 2′,5′-hydroxy-4-methoxychalcone has no natural precursor, its very similar to isoliquiritigenin
Isoliquiritigenin is a natural molecule of licorice recently shown to have antidiabetic, glucose-lowering effects (Gaur, 2013).
It’s also quite similar to the chalcone of isosakuranetin, the second most abundant grapefruit flavanone behind naringenin (Vanamala, 2004).
For this reason, phenolic grapefruit extracts could be more effective than pure synthetic naringenin gram-for-gram. Both are available online.
Isosakuranetin is nearly identical to naringenin, save for the methyl group, and differs from hesperetin by only one hydroxyl group.
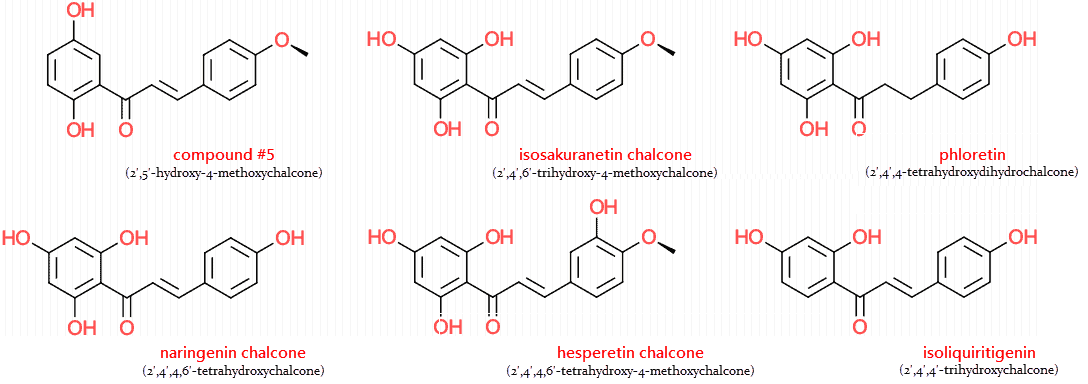
Phloretin is another antidiabetic chalcone found in apple skins.
It has recently been shown to induce adiponectin (Hassan, 2007), implying PPAR-γ activity, and is identical to naringenin chalcone save for two hydrogen atoms — i.e. one double bond.
The naringenin chalcone could be the “unidentified metabolite” uncovered in a chemical-distribution study on rats (Mohsen, 2009).
On account of there being more radioactivity in the tissues than could be explained by [H3]naringenin concentrations, the presence of uncharacterized breakdown products had been proposed:
“The contradiction between these reports and the results of the present study provide strong evidence for the existence of further unidentified metabolites of naringenin, which are either absorbed slowly or strongly retained by body organs.” ―Mohsen, 2009
It’s almost as if animals had evolved to sense flavanones, chalcones, and dihydrochalcones as signals of incoming glucose and respond accordingly.
There is no present consensus as to what the “natural ligands” of PPAR actually are, so anything they bind with can be proposed.
The presence of active METABOLITES of naringenin can be seen as extraneous considering the antidiabetic activity shown by the flavanone itself, yet it helps to explain why modest doses have been found so effective.
People need confidence that it’s not just “woo” or hype promulgated by Japanese scientists.
People should also know that they don’t necessarily need to take multiple grams of naringenin per day — though that should be safe.
There’s so many good in vivo rodent studies that’s it’s difficult to choose which one to use, but the one below uses realistic amounts of naringenin:

This study had used four groups of rats given naringenin at only 0, 0.003, 0.006, and 0.012% of diet.
After 6 weeks of diets also containing 16% fat and 45.5% sucrose, the fat rats were given a fatal dose of ketamine and then examined.
‘Thus, the dietary levels at 0.003, 0.006, and 0.012% naringenin supplementation employed in the current study would be equivalent to 1, 2, and 4 cups of grapefruit juice in humans.’ ―Cho, 2011
They initially found that naringenin caused dose-dependent effects on body weight, adipose tissue mass, and the triglyceride content of adipose tissue:
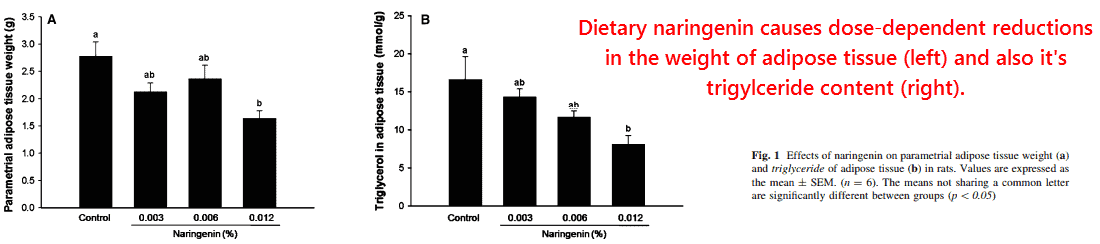
Plasma triglycerides also decreased in a dose-dependent manner: the concentrations of 86%, 51%, 50%, and 39% corresponded to increasing intakes of naringenin.
This lipid-lowering effect also happens with fibrates, which are classic PPAR-α treatments, and is perhaps why they chose to further examine the liver.
The liver is where PPAR-α is most highly expressed and the main site of plasma lipid clearance.
They found increased levels of carnitine palmitoyltransferase-1 and uncoupling protein-2, two proteins which increase the rate of fatty acid β-oxidation.
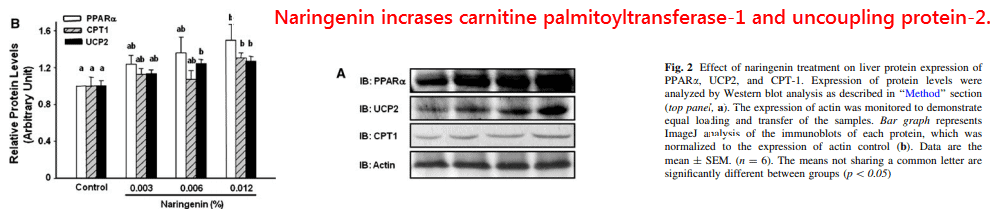
Other liver proteins upregulated by naringenin, but not by hesperetin, and transcribed through PPAR-α include the following:
Carnitine octanoyltransferase (188.6%), acyl-CoA oxidase (162.6%), and phosphofructokinase-2 (179.7%).
This other study used 1% naringenin, a substantial amount, and not surprisingly plasma triglyceride concentrations were reduced by nearly half (Huong, 2006).
The ability of PPAR-α agonists to lower plasma triglycerides is often used to explain their antidiabetic effects (Ferré, 2004).
But naringenin is unique in that it activates PPAR-γ as well.
As an agonist of both receptors, naringenin can improve the diabetic state by enhancing glucose flux in a variety of ways.
The increased expression of PPAR-γ genes adiponectin, uncoupling protein-1, glucose transporter-4, hexokinase, and pyruvate kinase have all been reported through naringenin-feeding.
Conversely, naringenin can also downregulate fat- and carbohydrate-producing enzymes such as glucose-6-phosphate dehydrogenase, fatty acid synthase, and fructose 1,6-bisphosphatase.
‘Our results showed that both blood and hepatic lipid levels were lowered by naringenin supplementation even at the lowest concentration of 0.003%.’ ―Cho, 2011
So even though naringenin is not the most POTENT PPAR-α or -γ agonist out there, it can activate both receptors so results are achievable at modest intakes.
But also, the known antidiabetic nature of its chalcone metabolites means it could be even more potent than what the cell transfection assays would indicate.
This is why studies like the one below are entirely believable:

Another thing making this study credible is the arrogant tone of the author.
He sounds like a person eager to dismiss the grapefruit diet as “a frivolous daydream of the laity” [paraphrase], and then uses the most simple and dismissive explanations when he found it to actually work.
Although Ken Fujioka is listed as lead author, I get the feeling that Frank Greenway, an American, wrote the bulk of it. Japanese people are generally more modest.
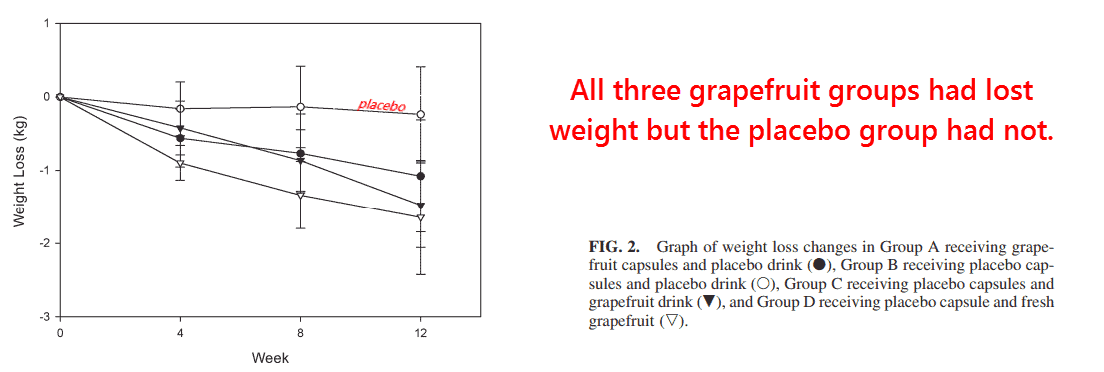
So they recruited over 100 people for this study and divided them into four groups:
(1) the grapefruit capsule group, (2) the placebo capsule group, (3) the 8-ounce grapefruit juice group, and (4) ½ whole grapefruit group.
All groups not taking grapefruit capsules were given placebo ones, and those not getting extra calories from grapefruit were given apple juice.
Everything was consumed three times per day regardless of group assignment.
Although apples have antidiabetic chalcones of similar efficacy to naringinen, these are contained primarily in the peel and found within the juice only in smallish amounts (DeLage, 1991).
This is approximately one-tenth the naringenin concentration found in grapefruit juice.
And the results do not disappoint. They are fully in accord with biochemical in vitro studies, nutritional animal studies, and anecdotal results of “grapefruit dieting.”
After three months, the placebo group remained essentially unchanged, yet all three grapefruit groups had lost weight:
There’s other evidence for naringenin besides.
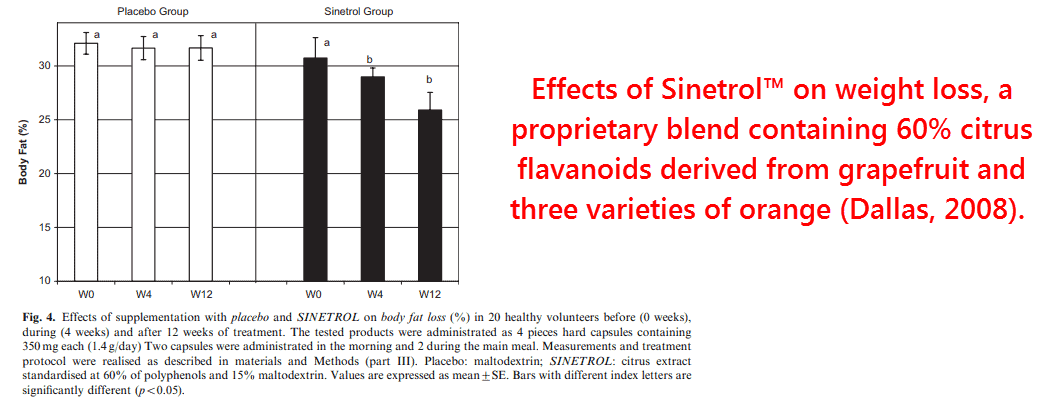
Two human studies using proprietary blends of citrus polyphenols reported reductions in weight and body fat percentage (Dallas, 2008; Dallas, 2014).
Another study employing bergamot polyphenols showed pronounced hypolipidemic and hypoglycemic effects (Mollace, 2010).
So grapefruit dieting is certainly not just anecdotal, and a person could safely take naringenin in higher amounts for quicker weight loss.
Naringenin and naringin are both available online without a doctor’s note, and can be obtained at a mere fraction of the cost of thiazolidinediones.
And since grapefruit extract also contains isosakuranetin, it could actually be even more effective than naringenin gram-for-gram…
‘This study gives credence to the lay public’s perception of grapefruit as a diet food and demonstrates that it has particular relevance to those with the metabolic syndrome […]’ ―Greenway, 2006
—-Important Message—-
Eat these foods at breakfast time — may reverse blood sugar symptoms in 2 weeks or less

Men eating these foods at breakfast time are finding a very nice testosterone increase…
And once blood sugars are normal, doctors can take them off their medications like Metformin, Cycloset, Avandia, Actos, Januvia, insulin, and more…
Even men who are prediabetic may find that their blood sugar goes from an average of 118 fasting down to 95 in just a few weeks…
Even type 2 diabetics and pre diabetics could possibly reverse their symptoms with this special breakfast.
And diabetic men — without taking any Big Pharma treatments — may report that their blood sugar goes from 300 or 200, down to 95 or 100.
Here’s the special breakfast men are eating that costs less than $1 to make
———-
