
This eliminates the opiates so you can eat what you want
—-Important Message From Our Sponsor—-
Vietnam vet discovers ancient jungle food that gives men INTENSE rockiness

He found it deep in the jungles of the Kontum region when he was fighting there.
And he noticed that whenever he ate this food, he got huge engorged erections and could last for hours.
Now today at quite an old age, he’s still using it to perform like a young man in bed.
Here it is — this strange Vietnamese secret found in the jungle…
———-
How to avoid opiates in food that cause constipation
Constipation is a well recognized side effect of painkillers and opiate addiction, yet the effect of opiate‑peptides derived from food (a.k.a. exorphins) is under‑recognized.
Of the four known exorphins only two of them, β‑casomorphin and soy morphin, interact with the μ‑receptor. This is the opiate receptor subtype found in the intestines responsible for motility.
The other two exorphins instead activate the δ‑opiate receptor and have less net effect on motility. Of these, gluten exorphin comes from wheat and rubiscolin derives from spinach.
Soymorphin naturally originates from soybeans and β‑casomorphin, the strongest exorphin, is produced exclusively from A2 casein.
Both opiate‑peptides have been shown to reduce intestinal motility, an effect reversible by naloxone in each case.
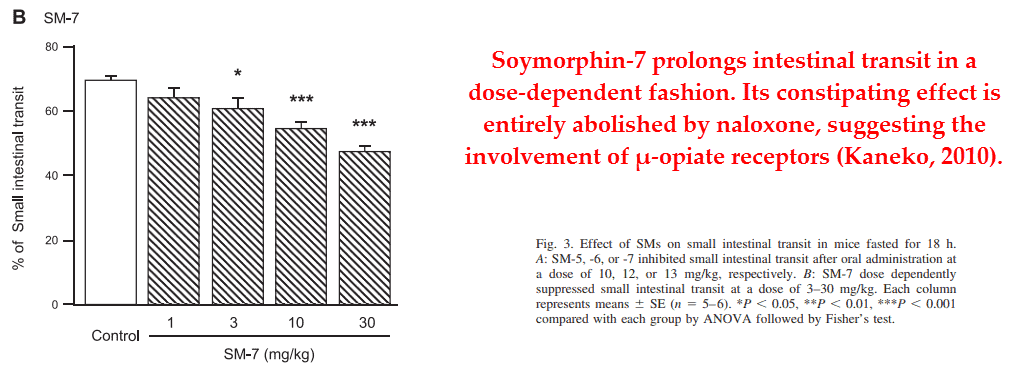
Naloxone is the classic anti‑opiate treatment and has been found effective in reversing idiopathic constipation, or constipation without any known cause.
Since naloxone has no effect on motility in the absence of opiates, this could imply that food‑derived exorphins commonly exert influence in humans.
Yet luckily, there are two known natural anti‑opiates: Caffeoyl quinide and lobeline.
Caffeoyl quinine is found in coffee beans and is formed upon roasting.
This natural molecule has been shown to displace [3H]naloxone from μ‑opiate receptors (Ki = 4.4 μM), reverse morphine anesthesia in rats, and is very likely what underlies the intestinal effects of coffee.
It’s not the caffeine because tea and soda don’t facilitate motility…
And the diterpenes kahweol and cafestol — which actually enhance bile flow — are excluded by the paper filter that absorbs it.
Lobeline is found in many plants but most notably Lobelia inflata, or “Indian tobacco.”
Lobeline is slightly more potent than caffeoyl quinide.
It displaces [3H]DAMGO from μ‑receptors at a concentration of .74 μM, and antagonizes morphine’s effect with an IC50 of 1.1μM.
The herbal usage of this plant is in accord with its pharmacology.
Lobelia inflata has been observed to reduce constipation for over a century.
‘Though sometimes overlooked when we are seeking a ‘treatment’ to overcome intestinal atony, experience has proved lobelia, continued for some time in moderately small doses, to be one of the best agents at our command to gradually relieve habitual constipation.’ –Felter, 1922
Although neither treatment is quite as powerful as naloxone — a classic synthetic — both are reasonably safe, effective, natural, affordable, and easily available.
And ostensibly, these are the only two natural anti‑opiates known to even exist.
Lobeline is a piperidine alkaloid, and the first anti‑opiate synthesized not based off morphine’s skeleton was an N‑substituted piperidine.
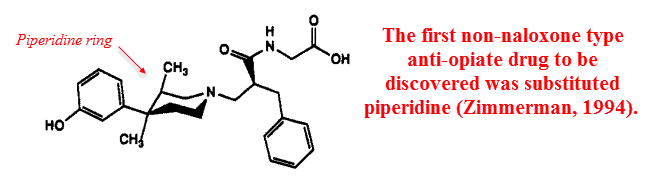
Since then, many synthetic opiates and anti‑opiates have been built around piperidine rings.
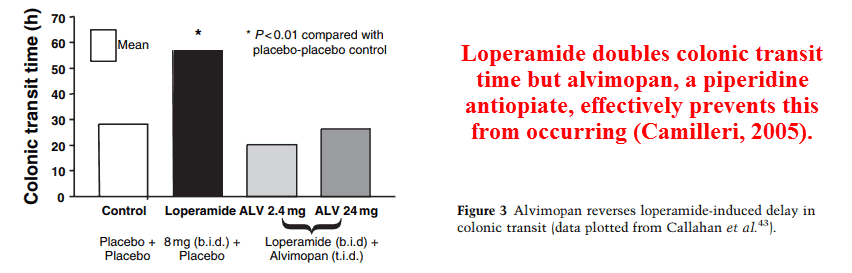
The two most well‑known piperidine agonists are loperamide and fentanyl — also a heroin adulterant — and the most popular antagonist is alvimopan, which is shown above.
Alvimopan is FDA‑approved for treating a specific form of postoperative intestinal paralysis, but is more commonly used off‑label for reversing constipation caused by painkillers.
And like naloxone, alvimopan has been shown to reduce constipation in subjects NOT taking opiates.
This is another indication that exorphins could unknowingly be exerting effects on people who eat them.
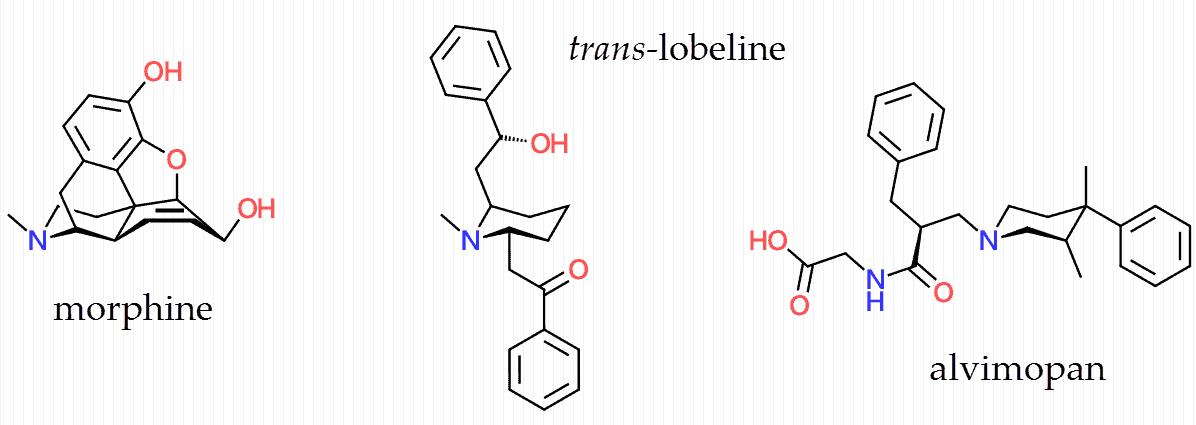
Lobeline is somewhat similar to alvimopan in that it features a piperidine ring, something the classic opiates also have buried within their structure.
This is similar to how an indole ring can be spotted within the larger polycycle of ergolines, such as LSD and bromocriptine.
It’s apparent that opiates are reliable constipation agents, and conversely: anti‑opiates are effective constipation relieving agents.
The most effective treatments of this class have piperidine ring structures, and piperine from black peppercorns is actually considered by some to reduce diarrhea by activating μ‑opiate receptors.
So when coffee alone is not enough, or objected to for whatever reason, extracts of Lobelia inflata could be a suitable choice.
Lobeline is somewhat analogous to alvimopan, albeit all‑natural and available without a doctor’s order.
Below is some evidence demonstrating lobeline’s antagonistic activity at μ‑receptors, and also a few reasons why it’s expected to be safer in humans than in rodents:

This study was the first to examine directly the anti‑opiate activity of lobeline.
Complementary RNA encoding opiate receptors had been injected into Xenopus oocytes, chosen on account of their size, leading to cells expressing high receptor density on their membranes.
In short, this is a very reliable method for ascertaining the effects of opiates.
Microelectrodes were then used to measure polarity changes induced by: (1) morphine, (2) the opiate‑peptide DAMGO, and (3) by these opiates in the presence of lobeline.
They had found lobeline to effectively antagonize morphine’s effect:

From this you could predict lobeline to reduce constipation similar to alvimopan, especially seeing as it’s historically known for doing so.
They had also determined its ability to displace [3H]DAMGO from guinea pig brains.
The opiate‑peptide DAMGO is similar to enkephalins, a class of opiates humans produce themselves, and is composed of five amino acids in a specific sequence (Tyr–Ala–Gly–Phe–Gly).
Enkephalins are high‑affinity ligands, and though lobeline wasn’t as potent as naltrexone it could displace [3H]DAMGO in low micromolar concentrations.
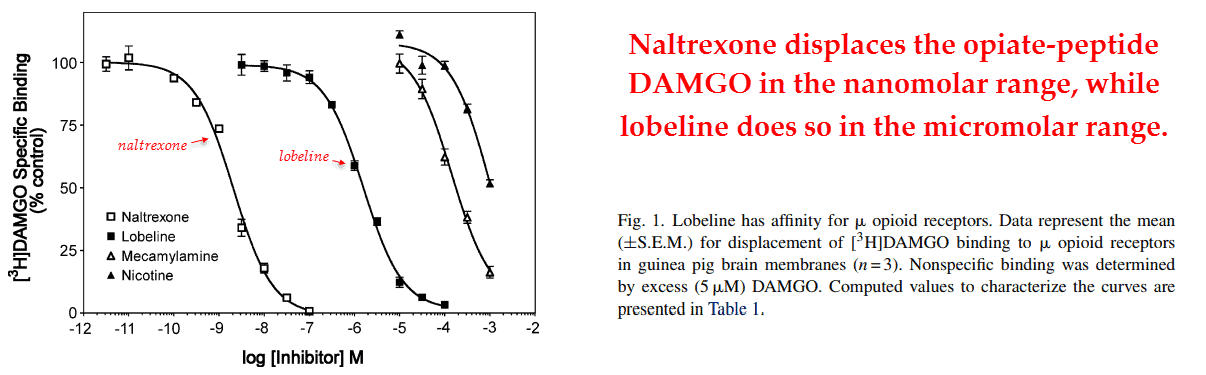
The potency of lobeline should be adequate for constipation, however, as it’s on the same order of magnitude as exorphins obtained from food.
Exorphins don’t bind μ‑receptors as strong as DAMGO, so concentrations of lobeline lower than .74 μM should effectively displace them.
Only a few hundred milligrams of dried Lobelia herb or a few drops of tincture would appear adequate for this purpose.
‘Rx Specific Medicine Lobelia, 1 or 2 drops, every 2 or 3 hours. This is accomplished by improving the innervation and peristalsis […]’ ―Felter, 1922
Doses up to 30 milligrams have been used clinically without excessive side‑effects, but the 7.5 milligram dose had most effectively improved cognition.
In smoking cessation trials, a 7.5 milligram dose has been taken nine times per day for six weeks thereby demonstrating its safety long‑term.
So what about lobeline’s other effects?
Lobeline is often used by people attempting to quit smoking because it’s historically been considered a cholinergic, similar to nicotine.
This would explain its ability to enhance cognition if true…
Yet studies indicate that the only acetylcholine receptor it has affinity for — the α4β2 subtype — is actually INHIBITED by lobeline in nanomolar concentrations.
Yet this shouldn’t matter after oral consumption in humans because lobeline enters the body in modified form.
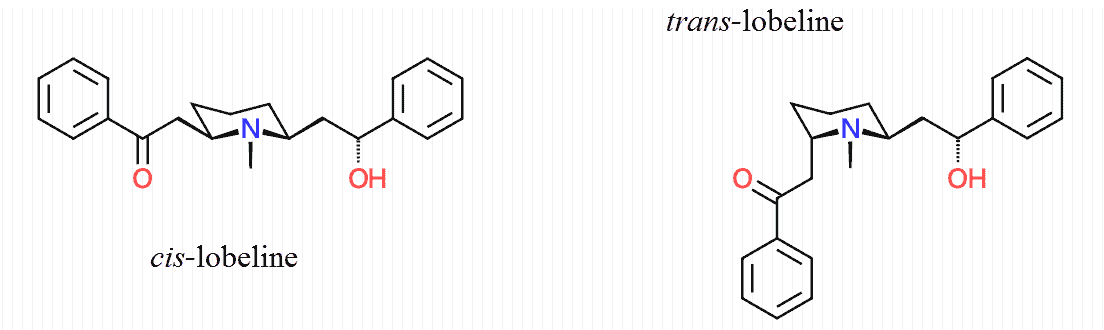
Only cis‑lobeline is ever used in binding assays because it’s the more potent epimer, yet data published a few years ago reveals that the trans‑epimer predominates in human plasma:
‘Interestingly, the cis∶trans lobeline ratio in human plasma after sublingual administration of lobeline sulfate is about 1∶19. Surprisingly, this ratio is reversed to 16∶1 cis:trans lobeline, in rat plasma after sublingual administration of lobeline sulfate.’ ―Crooks, 2011
This means that most in vitro binding data and activity studies are worthless, or all those except the ones investigating receptors found in external areas.
Cis‑lobeline could effectively reach the skin, intestinal tract, and respiratory tract when unmodified but becomes largely inactivated when taken orally.
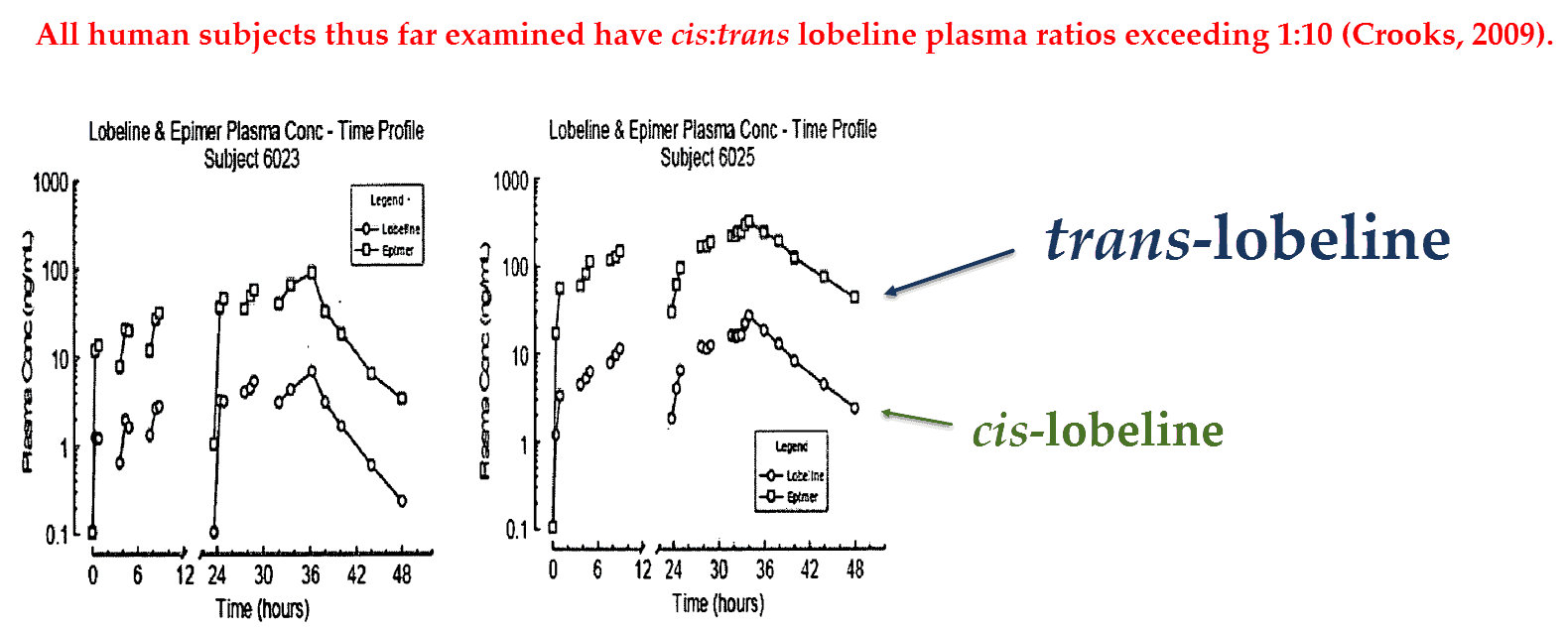
And considering that rodents demonstrate an inverted cis∶trans plasma ratio, all in vivo lobeline studies using rats are also worthless.
It’s been known for decades that lobeline exists in a more‑or‑less racemic form, or 50∶50 cis to trans, in solutions of pure water.
In rats, lobeline becomes more active because most is converted into the cis‑epimer upon assimilation.
Of course the converse is true in humans, a fact implying that toxicity studies in rodents could give a misleading impression of its safety.
Lobelia inflata has been used for centuries.
Renowned herbalists have attributed its inability to produce fatality in humans, but not animals, to its emetic effects being so pronounced that one would vomit before a toxic dose is reached.
Yet recently published data suggest its relative safety in humans could depend more on interspecies differences in pharmacokinetics than anything.
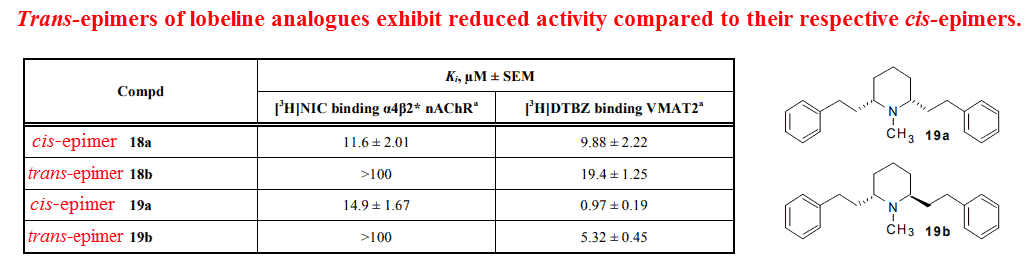
Although there’s scant data on the activity of trans‑lobeline explicitly on cellular targets, trans‑analogues of the molecule have been shown to display reduced affinity towards its classic binding sites — the α4β2 acetylcholine receptor and VMAT2, a monoamine transporter:
Brain uptake of trans‑lobeline has never been demonstrated, and considering it’s generally known to ENHANCE cognition I wouldn’t think α4β2 inhibition in vitro has any relevance.
Using 7.5 milligram doses given sublingually, lobeline has been shown to slightly increase cognitive test scores.
‘Under its action the mental powers are unusually acute, and the muscles are powerfully relaxed.’ —Felter, 1922
Unlike its binding to α4β2 acetylcholine receptors, which is completely abolished upon epimerization, trans‑analogues of lobeline still retain activity at VMAT2 — its other high affinity target.
Inhibiting VMAT2 is effectively preventing catecholamine uptake, perhaps the reason why lobeline has been shown effective in asthma.
The most common Rx treatments to treat this condition are actually catecholamine analogues, such as albuterol and salmeterol.
So in summary, lobeline can be included alongside caffeoyl quinide in the small list of natural anti‑opiates.
This treatment inhibits intestinal μ‑opiate receptors yet due to cis–trans epimerization, what happens once it gets inside the body cannot be inferred from extant data.
There’s simply not enough published data concerning trans‑lobeline, the primary circulating epimer in humans.
—-Important Message For Avoiding Endogenous Opioids—-
Beware these “natural” opioids that lead to tinnitus — ringing in the ears
I’ve discovered a way to stop endogenous opioids (EOs) from building up too high and causing problems in the body.
Because high levels of EOs lead to low T, fatigue, difficulty concentrating, memory loss, and a very annoying ringing noise in the ears.
It’s called tinnitus medically and it can get worse and worse…until it’s almost unbearable…
And it’s because of these endogenous opioids — they get too high and disrupt men’s natural opioid receptors in the body.
So here’s a solution — something that stops harmful EOs and boosts T in men
———-

Felter, Harvey. "The eclectic materia medica, pharmacology and therapeutics." Eclectic Medical Publications (1983) http://www.swsbm.com/FelterMM/Felters_Materia_Medica.pdf
Crooks, Peter. "Use of lobeline epimers in the treatment of central nervous system diseases, pathologies, and drug abuse." U.S. Patent Application No. 12/263,458 (2009) https://patentimages.storage.googleapis.com/3a/99/52/3277aa62bda041/US20090118331A1.pdf
Crooks, Peter. "Design, synthesis and interaction at the vesicular monoamine transporter‑2 of lobeline analogs: potential pharmacotherapies for the treatment of psychostimulant abuse." Current topics in medicinal chemistry (2011) https://www.ingentaconnect.com/content/ben/ctmc/2011/00000011/00000009/art00002
Martin, Catherine. "Lobeline effects on cognitive performance in adult ADHD." Journal of attention disorders (2018) https://www.researchgate.net/profile/Mark_Kleven/publication/256072510_Lobeline_Effects_on_Cognitive_Performance_in_Adult_ADHD/links/00b7d53c69dd5be4e4000000.pdf
Kaneko, Kentaro. "Orally administered soymorphins, soy‑derived opioid peptides, suppress feeding and intestinal transit via gut μ1‑receptor coupled to 5‑HT1A, D2, and GABAB systems." American Journal of Physiology-Gastrointestinal and Liver Physiology (2010) https://pdfs.semanticscholar.org/e227/13b22ec50491aad14220c392f7a9ec8cb54f.pdf
Miller, Dennis. "Lobeline, a potential pharmacotherapy for drug addiction, binds to μ‑opioid receptors and diminishes the effects of opioid receptor agonists." Drug and alcohol dependence (2007) https://www.sciencedirect.com/science/article/pii/S037687160700069
