
Here’s what you need to know before eating out
—-Important Message—-
This is Japan’s best male talent in “those” movies — and he’s 82 years old!
Meet 82-year-old Shigeo Tokuda, still going strong, making movie after movie…
…starring already in over 400 of “those” movies…

Find out what he is doing off camera to have such stamina at his age
———-
Hidden MSG in restaurant food
The late 1990s represented a period of monosodium glutamate (MSG) hysteria, or “monoglutamania” – where nearly everyone in the U.S. had reservations about consuming MSG.
The news apparently did not reach Japan, a country where MSG consumption remained at one kilogram per person per year throughout.
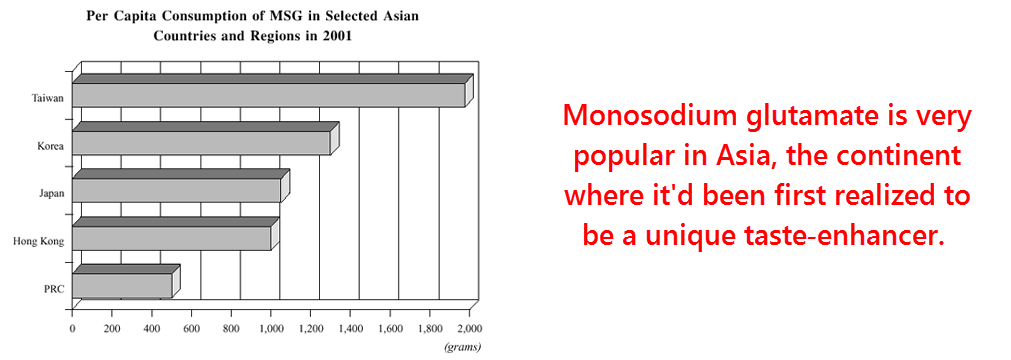
Nor did the reports seriously influence the Taiwanese, an Asian population who consumed – and still do – roughly twice that amount.
Although the roots of experimental glutamate excitotoxicity can be traced back to 1957, glutamate hysteria didn’t start in earnest until the late 1960s.
It was around that time that “Chinese restaurant syndrome” – a minor condition – was popularized by the press.
Yet Chinese restaurant syndrome displays symptoms more suggestive of added sulfites, not MSG.
Reports of Chinese restaurant syndrome sparked further research into glutamate excitotoxicity…
And the results were taken out of context and further sensationalized by the media.
Yet to this very day, excitotoxic brain lesions resulting from glutamate have never been shown to occur via the oral route.
Only injected doses of glutamate, and high ones at that, have ever been shown to cause trouble.
Moreover, excitotoxicity from glutamate only occurs in extremely young developing animals – and at concentrations far outside the physiological range.
In adult lab animals, the amounts of glutamate that must be injected to induce cell death are tenfold higher than in infants…
And those would be doses so high that calling them “heroic” would be an understatement.
“In mice, which have been studied more extensively for MSG-induced brain damage than other species, the lowest effective dose for the baby animal (0.5 g/kg) was approximately one-tenth that for the adult (5 g/kg).” (Olney, 1969)
That five grams per kilogram of body weight (5 g/kg) of MSG translates into 375 grams for a typical average-sized American (165 lbs).
As an extrapolated dose, it would also have to be injected…
And there’s simply no telling how much more it would take via oral consumption – because oral consumption has never been shown to induce excitotoxicity.
By comparison, a usual meal at any Chinese restaurant would rarely exceed 5 grams of MSG.
Excitotoxicity is also an artifact of in vitro cell studies (in vitro = in glass), where unphysiological concentrations of glutamate are used under artificial ionic conditions to kill neurons.
In addition, glutamate excitotoxicity has been shown to be strictly dependent on extracellular sodium (Na+) and calcium (Ca2+) concentrations (Choi, 1987).
And those are often higher in cell cultures due to “Earle’s balanced salt solution” being commonly used (Dubinsky, 1995).
Excitotoxicity occurs far less readily in vivo (in living cells) where cellular uptake mechanisms effectively keep those concentrations of Na+ and Ca2+ in range (Mangano, 1982).
Moreover, this process wasn’t known to be reversible until a few years before the turn of the century (Park, 1996)…
And that was decades after Chinese restaurant syndrome had irreparably damaged the reputation of MSG.
This means that most of the histological evidence of excitotoxicity originating from living animals (which only occurs through injection, remember) wasn’t even necessarily permanent.
Most microscopic evidence of excitotoxicity could have been the transient glutamate-induced swelling that results from the influx of sodium, chloride, and water into the cell.
In classic studies, animals had often been killed and studied very shortly after glutamate injection (Olney, 1969)…
Yet excitotoxicity is often reversible after a few hours (Choi, 1987).
“Serial observation of neurons exposed to glutamate in calcium-free solution showed that the resultant acute perikaryal (cell body of a neuron) swelling was short-lived…within the next hour the majority of these neurons recovered normal appearance and survived.” (Choi, 1987)
What conclusion does that bring us to?
Even if a person consumed more than 375 grams of MSG in one sitting – surprisingly, that’s only 1/3 of the median lethal dose – there’s a good chance they’d be fine in a few hours…
And that’s even if it initially caused “excitotoxicity.”
This may seem like old news to some, or that I am beating a dead horse.
Because many people are no longer afraid of MSG after being enlightened by the diversity of online information.
Only in times past could the mass media create such frenzies by monopolizing information.
Yet excitotoxicity is important nonetheless, at least for one particular reason…
Even though, for the most part, most people no longer have serious reservations about eating MSG, the concept of excitotoxicity may still subconsciously influence perceptions about glutaminergic drugs.
This could be unfortunate, because some of the most effective nootropic agents are agonists of the NMDA glutamate receptor…
That’s the very same receptor type most implicated in inducing excitotoxicity.
It almost seems like a paradox…
It’s an odd situation where the drugs found most clinically effective at increasing test scores are the same ones most expected to kill brain cells.
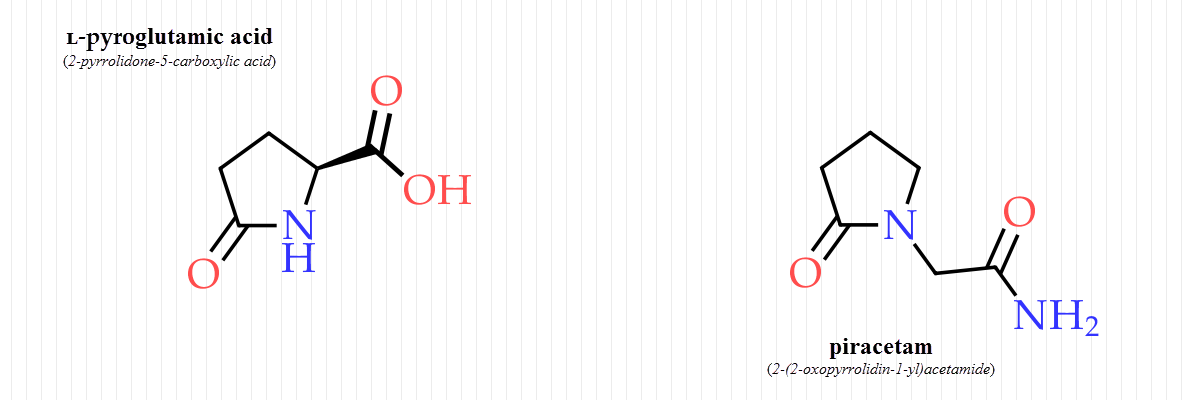
Piracetam, pyroglutamate, and D-serine have all been clinically shown to improve cognition in humans.
The latter two are completely natural amino acid metabolites shown to exist in the cerebrospinal fluid and plasma.
But that’s not to imply that monosodium glutamate should also enhance cognition…
This form of glutamate is ionized and largely incapable of crossing the blood-brain barrier (Schwerin, 1949).
This fact guarantees the safety of MSG in gram-sized doses, but it also excludes a nootropic role for it.
The only thing MSG enhances is taste… There is no evidence that it can significantly affect cognition one way or the other.
“…no study which used monosodium glutamate reported positive effects of the drug on intellectual functioning…” (Vogel, 1966)
On the other hand, the cyclic form of glutamate, L-pyroglutamic acid, freely enters the brain (Chanal, 1988).
A close cousin of pyroglutamate, piracetam, also crosses the blood-brain barrier.
This is the original “nootropic drug,” shown to improve cognition in countless studies.
So to benefit from these cognitive enhancers while being confident of their safety, the phenomenon of excitotoxicity must be put into perspective.
The science indicates that excitotoxicity does in fact happen, both in vitro and in young animals (through injection).
Yet it also suggests that it would be unlikely to occur under the doses commonly indicated for piracetam and L-pyroglutamate.
Unfortunately, the nootropic effects of these drugs cannot be separated from their excitotoxic potential at high doses.
And that’s because intracellular calcium is largely responsible for both effects.
The influx of calcium in neurons causes a rapid remodeling of the microtubule cytoskeleton (Forscher, 1989).
The microtubule cytoskeleton is the only cellular structure that can logically be thought to propagate signals at nerve impulse velocities.
Intracellular calcium has also been shown to enhance long‑term potentiation (Turner, 1982).
This is a phenomenon observed in the hippocampus whereby nerve tracts are strengthened upon repeated stimulation.
Glutaminergic drugs are classic regulators of long‑term potentiation…
And this effect could be an electrophysiological manifestation of learning itself.
Yet these same calcium ions are also what cause the delayed cell death seen with excessive concentrations of glutamate, kainate, and NMDA (N‑methyl‑ᴅ‑aspartate).

This study proved that glutamate excitotoxicity was strictly dependent on the concentrations of sodium, chloride, calcium, and magnesium in the extracellular fluid.
The study showed that sodium and chloride mediated the initial swelling phase, apparently the result sodium’s ability to draw in water.
The researchers proved this in a few ways, one being the use of hypertonic sucrose solution to counter osmotic effects.
They also used calcium and sodium-sensitive fluorescent indicators to quantify, via microscopy, the amount of the ions pulled into the cell.
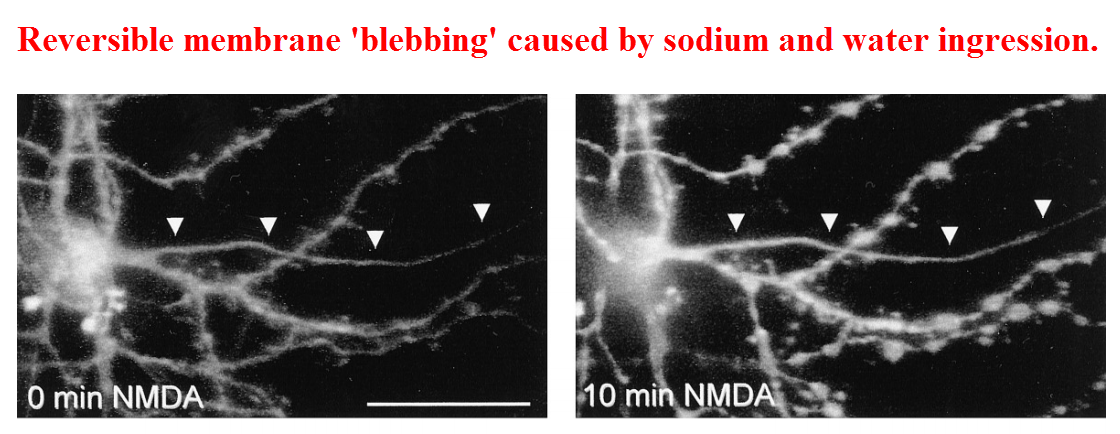
The sodium-dependent swelling was reversible. (As already mentioned, this was also noted eleven years prior.)
Yet the calcium admitted through the NMDA receptor was capable of permanently inducing cell death.
These ion-dependent effects are now fairly well established…
There are at least two other studies independent of each other that came to the very same conclusion.
“In dissociated cortical cultures, acute excitotoxic cell swelling after glutamate receptor activation is dependent on sodium and chloride entry, while mechanisms leading to delayed cell death are largely dependent on calcium.” (Hasbani, 1998)
According to Dr. Dennis Choi, the concentrations of glutamate needed to induce 40% cell death are roughly tenfold higher than the normal brain concentration.
Yet we already know that this is impossible to achieve in adult mammals…
Even 1.2 to 1.3 milligrams per kilogram of body weight had been shown incapable of increasing brain glutamate levels upon injection (Schwerin, 1949).
Moreover, it was shown that far higher concentrations are easily dealt with in vivo because of efficient glutamate uptake mechanisms in intact brains…
This effect is often overlooked when extrapolating in vitro results.
“The present data confirm earlier reports that both striatal and hippocampal tissues possess very efficient mechanisms to remove high amounts of endogenous excitatory amino acids from synaptic sites… Transport mechanisms could theoretically remove 20–32 μmoles (micromoles) of glutamate per minute.” (Mangano, 1982)
So glutamate certainly appears to be a non-issue… But what about piracetam and pyroglutamate?
These two molecules DO cross the blood-brain barrier.
But pyroglutamate can be exonerated from causing excitotoxicity, even at high doses.
This molecule has been shown to be innocuous even when fed orally at 4 grams per kilogram of body weight (for mice), which is 300 grams for a typical American:

This study used pyroglutamate in doses of 4 g/kg of body weight given to infant mice – the age where they are most vulnerable to intravenous glutamate.
They found absolutely no indication of excitotoxicity in the hypothalamus, the brain region classically known as the most influenced by excitotoxic agents.
In fact, one of the pyroglutamate groups (2 g/kg) ended up with less total necrotic neurons than the control mice that had been given nothing.
And the higher-dose pyroglutamate group (2 g/kg) had essentially the same number of total necrotic neurons than the controls (the subtle difference being 1.000 versus .909).

Much smaller doses of pyroglutamate have been shown to increase cognitive test scores in mice (e.g. 1.28 grams per day).
And since the mouse equivalent of a 300-gram oral dose for humans didn’t induce toxicity, it’s fair to assume its safety.
And this is despite this dose increasing brain levels 4.5-fold, proving yet again that pyroglutamate readily enters the brain.
And, incidentally, this research also proved that straight‑chain L-glutamate does not result in increased brain glutamate levels, confirming dozens of studies conducted before and since.
“In adult mice, plasma pyroglutamate levels were raised by a factor of 55.8 at 30 min whereas brain pyroglutamate was increased by a factor of 4.5.” (Caccia, 1983)
And, moreover, humans would be expected to have an even higher tolerance for pyroglutamate than rodents…
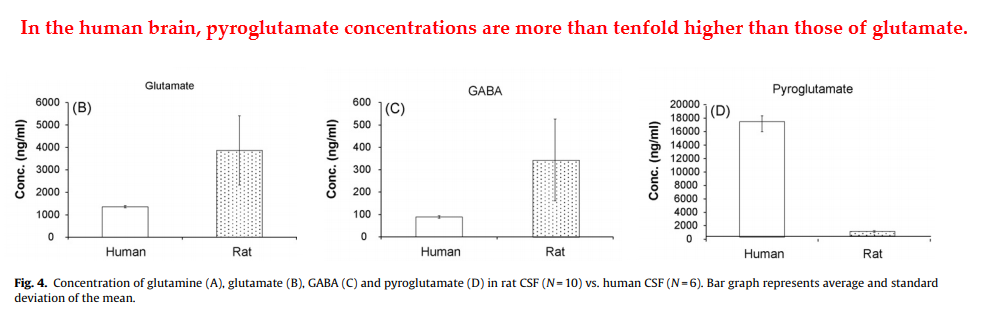
Unlike glutamate and gamma-aminobutyric acid (GABA), humans have more brain pyroglutamate compared to rats (Eckstein, 2008).
And because pyroglutamate is a completely normal metabolite, it shouldn’t come as a surprise that we have enzymes designed to efficiently metabolize it.
The enzyme pyroglutamate hydrolase (also called 5‑oxoprolinase or pyroglutamase) seems specific for this purpose.
And the product of this reaction is glutamate, a neurotransmitter associated with brain plasticity and learning – as long as its concentrations are kept within a reasonable range.
And since pyroglutamate is only a weak activator of the NMDA receptor, a glutamate precursor role could be what underlies its beneficial effects.
“In the central nervous system, systemic administration of pyroglutamic acid improves learning and memory in aged rats in behavioral studies.” (Oono, 2009)
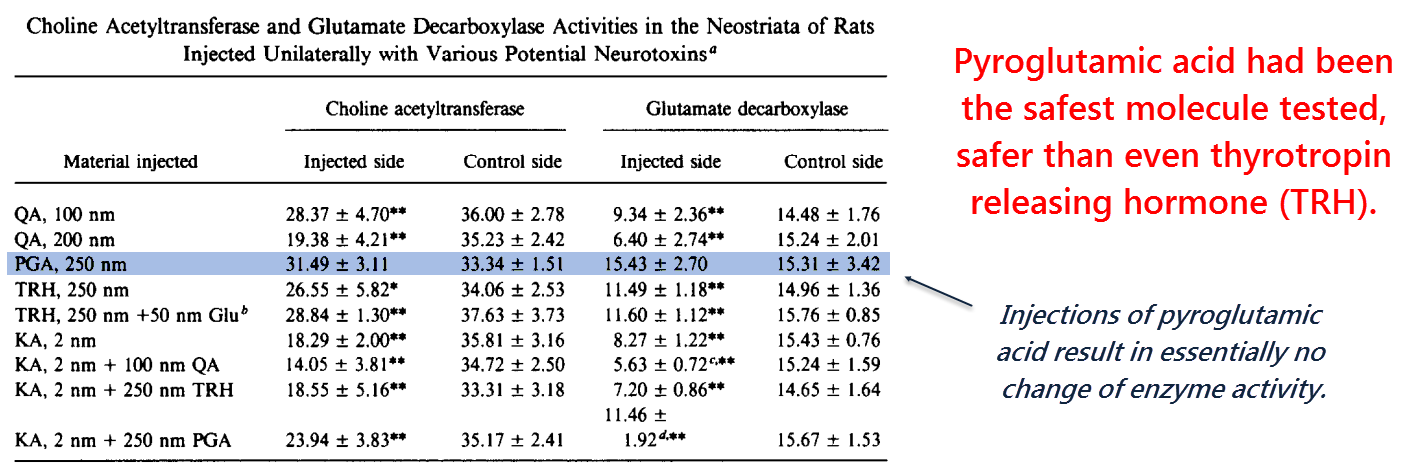
Pyroglutamate had been tested using direct brain injections under the suspicion it could be an endogenous excitotoxin.
But the results of that study showed it to be safe (McGeer, 1984).
The most potent endogenous excitotoxin turns out to be quinolinic acid, a tryptophan metabolite many times stronger than glutamate.
Thyrotropin‑releasing hormone (TRH) was also tested because it has an N‑terminal pyroglutamate residue.
In addition to the free form, the brain also contains pyroglutamate at the end of some peptide hormones.
Other studies do confirm this effect…
And pyroglutamate has even been considered “neuroprotective” by some.
Injections of 1% pyroglutamate solution have been shown to promote the survival of retinal cells in rats, after removal of the optic nerve (Oono, 2009).
Coincidentally, the retina is one of the classic target organs of excitotoxicity.
Over a half-century ago, injections of glutamate were shown to characteristically damage this area in developing rats (Lucas, 1957).
“A slight neurotoxicity was obtained with 250 nanomoles of thyroid-releasing hormone but not with a similar dose of L-pyroglutamic acid.” (McGeer, 1984)
So direct excitotoxicity is a non‑issue for pyroglutamate, a molecule that seems to gain its nootropic effects through other means.
Possibly it acts to improve cognition via being a glutamate precursor.
And/or also by inhibiting uptake, thereby keeping synaptic concentrations of glutamate high.
Regardless, pyroglutamate does have some activity on the NMDA receptor.
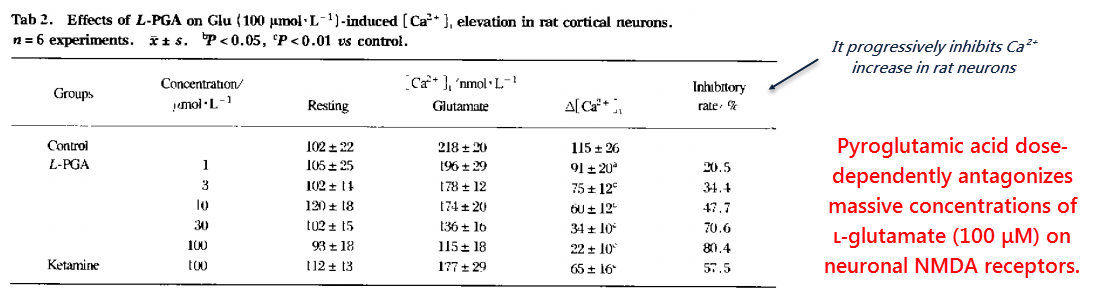
But that activity is weak enough that pyroglutamate effectively serves as an antagonist when competing against high concentrations of glutamate:

This study was of the usual in vitro type…
They used cultured rat neurons, impractical concentrations of glutamate, and unphysiological ion concentrations in the media they used.
They conducted this study using no magnesium (an ion shown to effectively antagonize NMDA‑induced calcium influx).
Without magnesium ions in the extracellular fluid, excitotoxic effects occurring through the NMDA receptor are accentuated.
“In contrast, cultures exposed to the same amount of glutamate in the presence of L-pyroglutamic acid appeared remarkably preserved…” (Xiao, 1999)
Nonetheless, pyroglutamic acid effectively antagonized glutamate in a dose-dependent manner.
This means that it’s weaker, and is yet another indication that it entirely lacks excitotoxic potential.
And yet, pyroglutamate has been consistently shown to improve cognition in a handful of human and animal studies.
Considering this fact, along with its low NMDA-receptor activity, it could be fair to view its effects as deriving from its glutamate precursor role.
The enzyme pyroglutamase has been shown to be expressed in the brain (Van der Werf, 1975).
And radiolabeled L-pyroglutamate yields increasing amounts of radiolabeled L-glutamate with time (Borg, 1986). (Radiolabeling is an investigative lab technique.)
Regardless of its exact mechanism, pyroglutamate seems to lack excitotoxic potential AND has a reputation for increasing cognition.
Since pyroglutamic acid is easily available online, it could be worth trying.
It is also naturally found in wine and cheese, and has been speculated to underlie the nootropic effects of Panax ginseng.
“L-pyroglutamic acid is one of the active components in several traditional Chinese medicines such as Panax ginseng and Ophiopogon japonicus.” (Xiao, 1999)
Piracetam is likewise free of any indication of excitotoxicity, and has actually been shown to promote the survival of neurons in many experiments (Brandão, 1995).
These two molecules are structurally similar, yet pyroglutamate has the benefit of being natural and available.
Because we humans have specific enzymes to regulate its concentration, long-term toxicity from ingesting pyroglutamate should be no concern.
And the list of experimental results derived from pyroglutamate studies reads somewhat like a piracetam article – which is not surprising considering their similarity in structure.
“L-pyroglutamic acid improved learning and memory capacity in aged rats, increased the blood flow and facilitated the uptake and utilization of glucose by brain tissue, exerted anxiolytic activity, antagonized the sodium glutamate-induced seizures in mice.” (Xiao, 1999)
—-Important Message—-
I’ve discovered what really causes Alzheimer’s disease — and how to naturally reverse the symptoms and keep a young mind and memory for decades longer
The way to appear young is to maintain a sharp memory and mind.
Because the moment you are forgetful is the moment those around you see you as “an old man.”
I’ve found that you can get and keep the sharpest memory and mind — never forget where you left your keys.
Never miss appointments. Remember what your boss needs from you and be the sharpest guy in the room.
Plus this discovery can even reverse Alzheimer’s symptoms.
I’ve dug up the studies that prove THIS is the true cause of Alzheimer’s, and I’ve also discovered a method to prevent it from ever happening to you.
So if you value your mind and your memory, and your dignity as a man, I urge you to watch this important video report and save yourself Alzheimer’s disease.
———-
